Mite pests in greenhouse crops: description, biology and management
Learn about mite pests, how to identify them, their life history and management strategies.
ISSN 1198-712X, Published May 2014
Introduction
Mites are more closely related to spiders than they are to insects. They are small (usually less than 1 mm in length), eight-legged organisms, with a wide host range, capable of causing extensive damage to a range of greenhouse crops. There are several species of concern. The most important is the twospotted spider mite (Tetranychus urticae); however, other species that can also cause significant damage include two other spider mites, the Lewis mite (Eotetranychus lewisi) and the carmine spider mite (Tetranychus cinnabarinus), as well as the broad mite (Polyphagotarsonemus latus), the cyclamen mite (Tarsonemus pallidus), the tomato russet mite (Aculops lycopersici) and the bulb mite (Rhizoglyphus spp).
Description and life history
Mites pass through a few stages from egg to adult. The eggs of the above-mentioned pests are laid singly on foliage or in the growing points of plants (or in the case of the bulb mite, in the soil).
Newly hatched mites pass through a six-legged larval stage and two eight-legged nymphal stages known respectively as the protonymph and deutonymph. The last of these is an immobile resting stage from which the adult emerges.
The twospotted spider mite (TSSM) (Figure 1) has a host range of hundreds of plant species, including all major vegetable crops and many ornamental crops. The eight-legged female adult is approximately 0.5 mm long, with a rounded abdomen. The male is distinguished from the female by its smaller, narrower body and pointed abdomen. Adults range from pale yellow to orange to brown or black. Day-lengths of 12 hr and less, decreasing temperatures and a deteriorating food source will induce diapause (an overwintering stage similar to hibernation).
The diapausing stage is reddish-orange in colour (Figure 2) and can tolerate very low temperatures. A short spell of heating is not sufficient to break diapause.
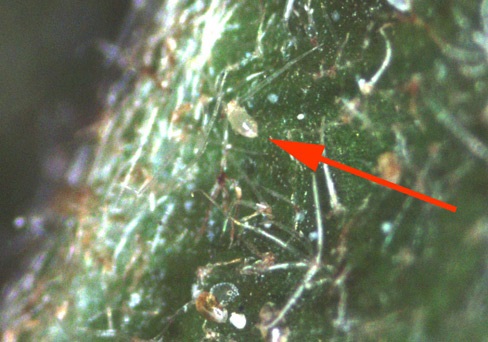
Close examination of leaf undersurfaces will show the mites to be miniscule moving dots. There are usually two dark spots on the body of TSSM, although this can be somewhat variable. After mating, each female mite lays approximately six pearly white eggs (Figure 3) a day. Over an average lifetime, a female lays 100 or more eggs on the undersurface of foliage. The life cycle from egg to adult ranges from 23 days at 15°C to just 4 days at 32°C. Development is fastest under hot, dry conditions.
The carmine spider mite and Lewis mite are closely related to TSSM. The carmine spider mite has not been positively identified in Ontario, but it is present in greenhouses in other countries. It is difficult to distinguish from TSSM in its immature stages, but the adult stage is bright red in colour and more commonly found on vegetable crops than in ornamentals. The Lewis mite is most often found on poinsettias and, in many ways, appears similar to TSSM, although it is often found with a few small spots on its back rather than the two distinctive spots of TSSM.
Broad mite (Figure 4) and cyclamen mite are microscopic in size (0.1–0.3 mm) and can be difficult to see even with a hand lens. For this reason, growers usually only know when they are present by the damage that they cause (see Damage). The life cycle (egg-to-adult) of these mites varies from less than a week under summer conditions to 10–18 days during winter, with the broad mite developing more quickly than the cyclamen mite.
These two mites can be difficult to distinguish from each other in the mobile stages. The most reliable method of identification is in the egg stage. Eggs are laid on the surface of young leaves (often along the mid-vein) or in developing flower buds or new growth. The broad mite egg has a very distinctive appearance, with small bumps covering the surface (Figure 5), compared with the cyclamen mite egg, which has a smooth surface (Figure 6).
Tomato russet mite belongs to a different family of mites known as gall mites and is even smaller than the broad mite and cyclamen mite. It is an elongated mite approximately 0.2 mm long and 0.05 mm wide (Figure 7). It differs from other mites in having only two pairs of legs in all stages. Because of its size, it often develops large populations before being noticed. It is primarily a pest of tomato but can also occasionally be found on other members of the tomato family.
Bulb mite (Figure 8), as the name suggests, can be a pest of crops such as lilies, tulips, gladiolus, daffodils and amaryllis. Slow moving, bulb mite has a white body, 0.5–1.0 mm in length, and short reddish-brown legs.
Damage
Mites feed by piercing the epidermis of the plant with sucking mouthparts and removing the cell contents. The various mite pests attack different crops and can be found on different parts of the plant.
Twospotted spider mite, Lewis mite and carmine spider mite are found primarily on the underside of leaves. The feeding injury caused by TSSM and Lewis mite is very similar and starts as a yellow "stippling" where the cell contents of the leaf have been removed (Figures 9 and 10). As mite numbers increase, the entire leaf appears stippled or light-coloured on the upper surface and develops a bronzed appearance under heavy mite populations. Very heavily infested leaves become yellow and brittle, and in some cases (for example, hibiscus), plants can exhibit a toxic response to feeding, with leaves yellowing and dropping from the plant even at quite low mite population densities.
If infestations proceed without control measures, plants may be killed. These mites produce webbing that is used by the mites to disperse with the aid of air currents. Large mite populations can produce large quantities of webbing (Figures 11 and 12, which can be unsightly (especially in ornamental crops). The mites also use the webbing as protection, increasing the difficulty of control with predators and with pesticides.
Carmine spider mite feeding can result in feeding damage that includes widespread leaf yellowing and leaf drop (Figure 13).
Broad mite and cyclamen mite each feed on a wide variety of crops and exhibit similar damage symptoms. They feed in the tight, newly emerging foliage and flower buds, with the damage becoming obvious as the plant tissue develops and enlarges. Toxins injected by the mites result in distorted, thickened and twisted growth at the top of the plant and in the flowers (Figure 14). The occurrence of these symptoms is usually the first indication that growers have a problem. These mites can be distributed throughout the greenhouse as hitchhikers on workers, equipment and even on insects such as whitefly (Figure 15).
Although there are subtle differences in the biology and damage symptoms between these two mites, they are often treated in the same way in terms of recognizing the problem and management strategies.
Tomato russet mite is another mite where the symptoms of damage are the first sign of the presence of the pest. They are found on the leaves, stem and fruit of tomatoes and, in large numbers, result in the plant tissue taking on a bronzed appearance (Figure 16). Damage symptoms include yellowing, curling and wilting of leaves, flower abortion and bronzed, cracked fruit. If uncontrolled, they will eventually kill the plant.
Bulb mites live and feed on plant parts below the soil surface. Feeding scars on the bulbs can turn brown and necrotic, and the wounds can create entry points for plant pathogens. The mites are attracted to and use damaged and diseased tissue to enter the bulb and feed. Often bulb mites and disease are found coexisting. Infested bulbs can show above-ground symptoms in the form of yellowing and stunting.
Management strategies
Spider mites
Good monitoring is critical to the early detection and management of spider mites. Pay close attention to new plant material entering the greenhouse and to susceptible crops and varieties. Monitor closely in areas of the greenhouse that are warmer and drier, for example, around heating pipes, south-facing walls and open vents/doorways. In ornamental crops, make regular crop inspections to detect early infestations before mite populations build up. In crops such as rose, inspect both upper and lower canopies. In poinsettias, Lewis mite may be introduced on cuttings. Inspect them carefully and monitor closely throughout the crop, paying attention to all varieties.
For vegetable crops, conduct a proper clean-up at the end of the crop to reduce initial infestations in the crop that follows. It would be better to reduce, if not eliminate, populations just before the overwintering or diapausing phase of the spider mites, since the diapausing mites hibernate in the ground, hollow stems, pipe fittings, cracks and crevices during the fall and winter. The mites become active again during late winter and early spring, and infestations in the new spring crop are often found where "hot spots" occurred during the previous fall. To detect infestations early, scouts should ideally check every row weekly. The red mite stages are generally pesticide resistant and are not as readily fed upon by predators. When the red diapausing mites are detected, use soap sprays on lightly infested leaves, and remove and destroy more heavily infested leaves.
Biological control
Spider mites can be controlled biologically using the predatory mite, Phytoseiulus persimilis. Other predatory mites used against this pest include some strains that are tolerant to high temperatures or pesticides. For example, the predatory mite Amblyseius californicus is reported to better tolerate dry conditions, while Amblyseius fallacis is resistant to some pesticides. Amblyseius andersoni is another predatory mite that has a wide range of temperature tolerances. Many Ontario growers have had good success with these predators.
Phytoseiulus persimilis — Phytoseiulus persimilis is about the same size as TSSM but is pear-shaped and pale salmon to bright orange (Figure 17). It also differs from TSSM in that it does not have two spots and moves more rapidly on long legs. It feeds on spider mites and does not diapause. Without spider mites, the predators die. This means new spider mite infestations require new introductions of the predator. Adult predators feed on about seven adults or 15–20 eggs per day. At 20°C, P. persimilis reproduces at almost twice the rate of TSSM. Control with P. persimilis is best between 20°C and 26°C. At temperatures above 30°C and humidity under 60%, the predators do not thrive and seek cooler, more protected areas lower in the crop canopy. In contrast, TSSM thrive under these conditions. The predators are available commercially either mixed with vermiculite, sawdust/wood chips or on bean leaves. With either carrier, treat infested plants at the first sign of damage. Try to place a few predators onto every infested leaf. Such placement is particularly important in crops (for example, tomato) with sticky hairs that interfere with the mobility of the predator between leaves. Before releasing the predators, ensure that they are alive and active.
Amblyseius californicus — Amblyseius californicus is a tan-coloured predatory mite similar in size to TSSM and P. persimilis. While it feeds primarily on spider mites, it can survive for longer in their absence than P. persimilis, feeding on other insects such as thrips, other mites and pollen. Its advantage over P. persimilis is that it develops more quickly at higher temperatures, remains active and effective at temperatures above 30°C and is less affected by low humidities. Amblyseius californicus can be used simultaneously with the more specialized P. persimilis, but this latter predator is best used by itself under low population conditions or in "hot spots," because A. californicus will also feed on P. persimilis.
Amblyseius fallacis — Similar in appearance to A. californicus, A. fallacis is a naturally occurring predatory mite in North American orchards. Likewise, it can survive in the absence of spider mites by feeding on other small insects and pollen. It has the benefit of remaining active and reproducing at lower temperatures than either P. persimilis or A. californicus. They can be used with other mite predators such as P. persimilis, Feltiella and Stethorus.
Feltiella acarisuga — Feltiella is a gall midge, a small predatory fly that lays its eggs on leaves infested by spider mites. When the larva (Figure 18) emerges from the eggs, it feeds on all stages of mites. The adult midge is not predatory. Feltiella enters diapause under short day conditions and as such is only suitable for use from March to September.
Stethorus punctillum — Stethorus is a small black ladybeetle (approximately 1.5 mm) (Figures 19 and 20) that feeds primarily on spider mites. Both adults and larvae are predatory. The adult is a good flier, locating spider mite infestations in the crops and laying eggs within the colony. This predator may not thrive on some host plants that have sticky hairs (for example, tomato).
Cultural control
Misting plants and raising the humidity will help suppress spider mite populations. For example, at 20°C and 36% relative humidity, a female TSSM will lay about seven eggs per day, while at 95% humidity, approximately 30% fewer eggs are laid.
Chemical control
Because of their great reproductive potential, TSSM can quickly develop pesticide resistance. To effectively manage this pest using pesticides, observe these guidelines:
- Direct sprays to the underside of leaves where spider mites usually congregate.
- Be sure to achieve good coverage, particularly when using contact miticides such as Dyno-Mite, Floramite and Shuttle.
- Use higher spray pressures to penetrate the web in areas of high mite density to reach the mites and the eggs within and beneath the web.
- Use non-chemical control options as much as possible to minimize the development of pesticide resistance.
Broad mite/cyclamen mite
Observation of damage is usually the first indication of the presence of these mites. Know what the damage looks like and which crops are most susceptible to damage. Educate greenhouse workers so that any suspect plants are quickly brought to the attention of the grower or IPM manager. Early removal of infested plants or plant parts and good weed control inside and outside the greenhouse can slow the establishment and spread of these mites.
Various predatory mites are reported to feed on these mites, including Neoseiulus cucumeris, Amblyseius swirskii, A. californicus and A. andersoni. Neoseiulus cucumeris, A. californicus and A. swirskii have demonstrated effective suppression of broad mites on crops such as peppers and begonia in greenhouse studies. As with many pests, however, it is likely to be much more effective when the predatory mites are already in the crop prior to any infestation developing. If using biocontrol, apply higher numbers of predators in areas where damage is observed.
Pesticides can control broad mites and cyclamen mites, but not many pesticides are registered for their control in Canada and there is the potential for compromising biocontrol of other pests. Use pesticides with caution and check with the side-effects lists at sites such as Koppert and Biobest.
Tomato russet mite
As with broad mite and cyclamen mite, the first indication that growers usually have of their presence is when damage is observed. The mites disperse though the greenhouse on the hands, clothing and equipment of workers. When detected, take appropriate measures that could include removing plants or affected plant parts.
Predatory mites such as A. fallacis and A. swirskii may have some potential for managing tomato russet mite. One Ontario study indicated that several releases of high populations of A. swirskii on tomato plants, particularly at the "leading edge" of tomato russet mite infestations, can suppress spread of this mite on individual plants. However, it may be best to adopt an integrated approach that includes monitoring, registered biocompatible pesticides and releases of predatory mites on plants showing symptoms.
Ensure that there are no potential host plants in the greenhouse between crops. A thorough clean-up inside and outside the greenhouse will help reduce incidence and spread.
Bulb mite
Bulb and root rot diseases are often found in association with bulb mites, either as a pre-cursor to the mite or as opportunistic invaders taking advantage of mite feeding to enter the plant. As such, fungicide treatment may be appropriate to minimize the impact of disease. Biological control of mites has been demonstrated with the predatory mite Gaeolaelaps (=Hypoaspis) aculeifer. Other generalist soil-dwelling predators, for example, the rove beetle Dalotia (=Atheta) coriaria, may also offer some control. However, for short-term ornamental crops such as tulips, narcissus and amaryllis, if the bulbs are heavily infested, the crop cycle may be too short for biocontrol to be effective. This reinforces the need for careful monitoring of the bulbs when they first arrive.