Downy mildew in cucurbits
Learn about downy mildew signs and symptoms and management strategies for this disease in cucurbit crops. This technical information is for commercial cucurbit producers in Ontario.
ISSN 1198-712X, Published July 2020
Introduction
This factsheet provides information on cucurbit downy mildew to better manage this disease in cucurbit crops in Ontario.
Cucurbit downy mildew, caused by the oomycete Pseudoperonospora cubensis, is a serious disease of cucurbit crops in Ontario (Figure 1). Once established in a region, the pathogen can rapidly spread through airborne inoculum, leading to severe defoliation, significant yield loss, decreased crop quality and plant death. Pseudoperonospora cubensis infects gourds, squash, pumpkins, melons and cucumber. Of these, cucumbers are the most susceptible to infection. During favourable environmental conditions, entire fields can be destroyed within a week as a result of severe defoliation.

Figure 1. Cucumber crop severely infected with downy mildew, leading to sunscald on the fruit.
Symptoms and signs
Downy mildew symptoms first appear as water‑soaked lesions on the topside of leaves. The lesions can start anywhere in the canopy. These initial lesions are best seen during a heavy dew. The centre of the lesion then turns yellow (chlorotic) and eventually tan or brown (necrotic) (Figure 2, top left). In cucumbers, lesions are often confined by leaf veins and appear angular in shape. In gourds, squash, pumpkins and cantaloupe, lesions appear irregularly shaped (Figure 2, top right). On watermelon leaves, the lesions tend to be smaller and rounder (Figure 2, bottom). As the disease progresses, lesions expand and multiply, causing foliage to become necrotic, and leading to plant death.

Figure 2. Downy mildew on cucumber (top left), cantaloupe (top right) and watermelon (bottom).
Under humid conditions, a downy growth that resembles “dirt” often develops on the underside of the initial water-soaked lesions. This growth on the underside of the leaf can also be seen before any symptoms on the upper leaf (Figure 3). This downy growth is particularly noticeable in the morning after a period of wet weather or when conditions favour dew formation.

Figure 3. Black growth on underside of the leaf, this is best seen during the early stages of infection. Upper leaf may not have noticeable lesions.
The downy growth on the underside of the lesions consists of black sporangiophores that contain the sporangia. These structures are visible with a 10x to 20x hand lens (loupe). The presence of downy growth on the underside of the lesion is a key diagnostic feature of this disease. Under a compound microscope, the lemon-shaped sporangia can be seen (Figure 4).
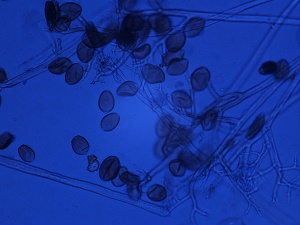
Figure 4. Sporangia and sporangiophores of Pseudoperonospora cubensis under a compound microscope. Photo courtesy of Dr. Shannon Shan, Pest Diagnostic Clinic, University of Guelph.
Due to the rapid spread of this disease and because symptoms often do not appear until 5–7 days after infection, a successful management program must be implemented prior to the appearance of the symptoms.
Downy mildew vs. angular leaf spot
Downy mildew can be commonly confused with angular leaf spot (Figure 5). Angular leaf spot is caused by the bacteria Pseudomonas syringae pv. lachrymans. It is important to distinguish between the two, as the management strategies are different. Angular leaf spot will not have black downy growth on the underside of the leaf.

Figure 5. Angular leaf spot caused by bacteria on butternut squash. Note the lack of black growth from the sporangiophores on the underside.
Pathogen biology
Downy mildew development is favoured by cool, wet and humid conditions. The pathogen produces microscopic sac-like structures called sporangia over a wide range of temperatures (5°C–30°C), however optimum reproduction occurs from 15°C–20°C. The longer the leaf wetness period, the higher the chances of infection over a larger range of temperatures. The minimum leaf wetness period is only 45 minutes. The sporangia are easily transferred to healthy plant tissue by air currents. Once they land on a susceptible host and have a prolonged wet period, the sporangia germinate and release zoospores. The zoospores swim through the film of water along the leaf surface towards the stomates. These natural pores are a primary point of entry for the pathogen, resulting in multiple infections on the leaf.
Infection from P. cubensis may progress slowly or stop temporarily when temperatures rise above 30°C during the day. However, nighttime temperatures of 12°C–23°C promote disease development, especially when accompanied by heavy dews, fog or precipitation. With nighttime temperatures around 15°C and daytime temperatures around 25°C, downy mildew infections on cucurbits produce more sporangia within 5 days.
Recently, there have been two host-specific clades identified for this pathogen. Clade I primarily infects pumpkin, squash and zucchini, and Clade II primarily infects cucumbers and cantaloupe. Prior to this, it was thought that there were six pathotypes associated with the different host ranges. It is suspected that Clade II is more aggressive than Clade I and that Clade II is often associated more with fungicide insensitivity (see Fungicide Resistance Management).
Pathogen survival and spread
The downy mildew pathogen is an obligate parasite. It requires living green plant tissue to survive. Killing frosts and cold winters effectively prevent spores from overwintering in the field in Ontario. However, downy mildew can overwinter on living cucurbit plant material growing in greenhouses. Furthermore, greenhouse cucumber crops, cucurbits grown in high tunnels and transplants are at risk of developing downy mildew from wind‑borne sources early in the spring, before the field crop has been planted.
Downy mildew primarily overwinters in the southern U.S. and Mexico, where cucurbits are produced year-round. In these areas, the inoculum builds up on susceptible hosts in the early spring. Sporangia are carried long distances by storms and may survive for several days. Greenhouses may also be providing initial inoculum, but the role is still unknown. Once the pathogen arrives in Ontario and becomes established, sporangia are spread through the air.
The Cucurbit Downy Mildew Forecast is a web-based downy mildew forecasting system that follows the movement of downy mildew from the south to north throughout the growing season and alerts growers to the potential movement of the disease into a region. Following the movement of the disease throughout the growing season and adhering to the regional disease alerts allows growers to make timely fungicide applications.
Management strategies
Use an integrated approach to manage cucurbit downy mildew. Downy mildew is a community disease, and a coordinated management strategy is the best defence against economic losses.
- If possible, produce vegetable transplants in greenhouses dedicated solely to transplant production. Do not produce cucurbit transplants in the same greenhouse as mature greenhouse cucumber plants.
- When planting cucurbit transplants, ensure that the transplants are free from disease.
- Where possible, use resistant or tolerant varieties.
- Select fields and manage the crop to promote air movement and reduce humidity levels inside the crop canopy.
- Avoid excess overhead irrigation. Consider irrigating during the late morning to facilitate rapid leaf drying. Apply a preventive fungicide prior to an overhead irrigation event. If possible, use trickle irrigation.
- Scout fields for symptoms of the disease every week — more often when conditions are favourable.
- Maintain good weed control in the field. Control alternate weed hosts for downy mildew, including wild cucumber, goldencreeper and volunteer cucurbits, in neighbouring fence rows and field edges.
- Follow a preventive spray program. Under high risk conditions, apply fungicides on a weekly rotation. Ensure adequate coverage and spray penetration into the canopy. Follow fungicide resistance management guidelines. For specific fungicide recommendations, see Ontario Ministry of Agriculture, Food and Rural Affairs (OMAFRA) Publication 838, Vegetable Crop Protection Guide and ONvegetables.com.
- Monitor the Cucurbit Downy Mildew Forecast website and local scouting reports to follow the movement of the disease throughout the growing season and make timely fungicide applications.
Fungicide resistance management
Fungicides can be an important tool in managing the impact of downy mildew on cucurbit crops. Resistance management needs to be followed for all downy mildew–specific fungicides. Management strategies include mixing the downy mildew–specific fungicides with low risk, preventive fungicides. Rotating fungicide groups (modes of action) is also important for resistance management. These strategies reduce the selection for resistance on the targeted fungicide, which is at high risk for resistance. Additionally, keeping the cucumber crop as healthy as possible and using other management strategies can help reduce reliance on fungicides.
Additional web resources
- Forecasting website for North America
- Michigan State University
- Publication 838, Vegetable Crop Protection Guide
References
Agrios, G.N. (2005). Plant Pathology. 5th ed. pp. 427–433.
Crandall, S.G., Rahman, A., Quesada-Ocampo, L.M., Martin, F.N., Bilodeau, G.J., & Miles, T.D. (2018). Advances in diagnostics of downy mildews: lessons learned from other oomycetes and future challenges. Plant disease, 102(2), 265–275.
Keinath, A.P., Wintermantel, W.M., & Zitter, T.A. (Eds.). (2017). Compendium of Cucurbit Diseases and Pests. St. Paul, MN: APS Press.
Savory, E.A., Granke, L.L., Quesada-Ocampo, L.M., Varbanova, M., Hausbeck, M.K., & Day, B. (2011). The cucurbit downy mildew pathogen Pseudoperonospora cubensis. Molecular plant pathology, 12(3), 217–226.
This factsheet was updated by Katie Goldenhar, Pathologist – Horticulture, OMAFRA, Guelph, and Andrew C. Wylie, Acting Vegetable Crop Specialist, OMAFRA, Ridgetown. The factsheet was originally written by Michael Celetti and Elaine Roddy, OMAFRA.