Shumard Oak Management Plan
This document advises the ministry on ways to ensure healthy numbers of Shumard Oak, a species of special concern, return to Ontario.
Management plan prepared under the Endangered Species Act, 2007
June 2013
About the Ontario Management Plan Series
This series presents the collection of management plans that are written for the Province of Ontario and contain possible approaches to manage species of special concern in Ontario. The Province ensures the preparation of the management plans meet its commitments to manage species of special concern under the Endangered Species Act, 2007 (ESA, 2007) and the Accord for the Protection of Species at Risk in Canada.
What is a species of special concern?
A species is classified as special concern if it lives in the wild in Ontario, is not endangered or threatened, but may become threatened or endangered due to a combination of biological characteristics and identified threats.
What is a management plan?
Under the ESA, 2007, a management plan identifies actions that could be taken to ensure, at a minimum, that a species of special concern does not become threatened or endangered. The plan provides detailed information about the current species population and distribution, their habitat requirements and areas of vulnerability. The plan also identifies threats to the species and sets a clear goal, possible strategies, and prioritized activities needed to address the threats.
Management plans are required to be prepared for species of special concern no later than five years of the species being added to the Species at Risk in Ontario list as a special concern species.
What’s next?
Nine months after the completion of a management plan a government response statement will be published which summarizes the actions that the Government of Ontario intends to take in response to the plan and the government priorities in taking those actions. The implementation of the management plan depends on the continued cooperation and actions of various sectors, government agencies, communities, conservation organisations, land owners, and individuals.
For more information
To learn more about species of special concern in Ontario, please visit the Ministry of Natural Resources Species at Risk webpage at: www.ontario.ca/speciesatrisk
Cover photo credit: Robert H. Mohlenbrock @ USDA-NRCS PLANTS Database / USDA SCS. 1989. Midwest wetland flora: Field office illustrated guide to plant species. Midwest National Technical Center, Lincoln.
Recommended citation
Donley, R.N., J. Jalava, and J. van Overbeeke. 2013. Management Plan for the Shumard Oak (Quercus shumardii) in Ontario. Ontario Management Plan Series. Prepared for the Ontario Ministry of Natural Resources, Peterborough, Ontario. v + 59 pp.
Cover illustration: Robert H. Mohlenbrock @ USDA-NRCS PLANTS Database / USDA SCS. 1989. Midwest wetland flora: Field office illustrated guide to plant species. Midwest National Technical Center, Lincoln.
© Queen’s Printer for Ontario, 2013
ISBN 978-1-4606-2032-8 (PDF)
Content (excluding the cover illustration) may be used without permission, with appropriate credit to the source.
Cette publication hautement spécialisée « Management plans prepared under the Endangered Species Act, 2007 », n'est disponible qu'en anglais en vertu du Règlement 411/97 qui en exempte l'application de la Loi sur les services en français. Pour obtenir de l'aide en français, veuillez communiquer avec le ministère des Richesses naturelles au 1-800-667-1940.
Authors
Rhonda Donley, Carolinian Canada Coalition
Jarmo Jalava, Carolinian Canada Coalition
Jennifer van Overbeeke, Carolinian Canada Coalition
Acknowledgments
The authors wish to acknowledge and thank the following people and organizations for their assistance: Daria Koscinski, Carolinian Canada Coalition, produced the map. Ben Porchuk contacted First Nations for Aboriginal Traditional Knowledge and updated information on relevant occurrences of Shumard Oak. The Natural Heritage Information Centre provided access to their paper files and occurrence data. Gerry Waldron reviewed parts of the draft and shared his wealth of knowledge on the species. Tim Payne provided access to St Clair Region Conservation Authority documents and answered questions about forest management. Mary Gartshore provided information on the St. Williams Forestry and Ecology Station’s nursery operations and on threats to the species. Craig Willet shared information on the Wheatley Woods Native Plant Nursery’s history with Shumard Oak. Robert Davies provided detailed information on Essex Region Conservation Authority’s Shumard Oak seed collection, planting activities and silvicultural methods for managing oaks. Chris Zoetewey answered questions about vegetation monitoring, deer impacts and deer herd management on Navy Island. John Ambrose, Karine Beriault, Graham Buck, Eric Cleland, Albert Garofalo, Brett Groves, Ron Ludolph, Alistair MacKenzie, Terry Schwan, Randall Van Wagner (Lower Thames Valley Conservation Authority), Tony Zammit (Grand River Conservation Authority) and Allen Woodliffe contributed valuable knowledge and information. Maria Kuzmina and Jeremy deWaard (Biodiversity Institute of Ontario) provided information on the Barcode of Life project collections. Tammy Dobbie, Dwight Bourdin, Teresa Austrin and Graham Buck provided contact details for some of the authorities consulted.
Declaration
The management plan for the Shumard Oak was developed in accordance with the requirements of the Endangered Species Act, 2007 (ESA). This management plan has been prepared for the Government of Ontario, other responsible jurisdictions and for the many different constituencies that may be involved in managing the species.
The management plan does not necessarily represent the views of all of the individuals who contributed to its preparation, or the official positions of the organizations with which the individuals are associated.
The goals, objectives and management approaches identified in the plan are based on the best available knowledge and are subject to revision as new information becomes available. Implementation of this plan is subject to appropriations, priorities and budgetary constraints of the participating jurisdictions and organizations.
Success in the management of this species depends on the commitment and cooperation of many different constituencies that will be involved in implementing the directions set out in this plan.
Responsible jurisdictions
Ontario Ministry of Natural Resources
Parks Canada Agency
Department of National Defence
Environment Canada – Canadian Wildlife Service, Ontario
Executive summary
Shumard Oak is a large tree with deeply-furrowed bark, massive limbs, a heavily- buttressed trunk and an open, spreading crown. Its leaves are shiny and have deep indents or spaces (sinuses) between the lobes of the leaf, helping to distinguish it from the similar-looking Red Oak. The autumn leaves of Shumard Oak are deep crimson or wine red colour and remain on the tree longer than other native oaks. Because of its very low population size in Ontario and threats to its habitat and individual trees, Shumard Oak was designated as special concern by the Committee on the Status of Endangered Wildlife In Canada (COSEWIC) in 1984 (reassessed in 1999), and by the Ontario Ministry of Natural Resources in 1988.
Shumard Oak is native to eastern North America and in Canada is restricted to the Carolinian life zone in extreme southern Ontario in Essex, Lambton, and Elgin counties and the regional municipalities of Chatham-Kent and Niagara. It grows on moist, well-drained loamy soils of stream and river terraces, and adjacent ridges and bluffs, as well as on moist slopes and poorly drained uplands. There are only 40 to 50 known locations for the species in Ontario. Many current and historical locations are of single, relict trees along fencerows and roads or in fields. Some populations are in protected areas and some show evidence of regeneration.
Shumard Oak faces a number of threats in Ontario including:
- habitat loss due to development, and woodlot and hedgerow removal to expand farming operations;
- habitat degradation caused by livestock grazing, human-caused hydrological changes and invasive species;
- mortality caused by a wide range of insect pests and diseases;
- inadvertent cutting of or damage to trees because of identification difficulties;
- herbivory of acorns by over-abundant White-tailed Deer; and
- harvest of acorns and trees for human uses.
This management plan provides approaches that could be taken to maintain or enhance self-sustaining populations of Shumard Oak within its current range in Ontario. The management actions are grouped under the following objectives:
- Protect and manage habitat to maintain extant populations in Ontario.
- Support efforts to research and control the spread of diseases and pests considered to be high-priority threats to Shumard Oak.
- Build capacity to accurately identify Shumard Oak.
- Determine the current distribution and abundance of Shumard Oak populations in Ontario through inventory and monitoring.
- Prepare and disseminate information on best management practices for Shumard Oak.
- Support and implement landscape- and ecosystem-based planning and recovery initiatives to increase the amount of available habitat for Shumard Oak, and to enhance habitat connectivity for dispersal and population expansion.
1.0 Species assessment and classification
Common name: (population): Shumard Oak
Scientific name: Quercus shumardii Buckl.
SARO List Classification: Special Concern
SARO List History: Special Concern (2004)
COSEWIC Assessment History: Special Concern (1984), reassessed (1999)
SARA Schedule 1: N/A (SARA Schedule 3: Special Concern)
Conservation status rankings: GRANK: G5 NRANK: N3 SRANK: S3
The glossary provides definitions for the abbreviations above.
2.0 Species information
2.1 Species description and biology
Species description
Shumard Oak (Quercus shumardii) is a large tree (up to 40m tall) with deeply-furrowed bark, massive limbs, a heavily-buttressed trunk, and an open, spreading crown. Shumard Oak is a member of the Lobatae section of the Quercus genus (eFloras 2008). The trees in this section are sometimes called "black oaks" but are more commonly referred to as the "red oaks". Shumard Oak and related "red oaks" can be difficult to identify because they readily hybridize. For this reason a detailed description is provided below and further in Appendix 1.
Mature bark of Shumard Oak is thick, grey-brown to dark brown, with pale ridges separated by dark shallow fissures. The inner bark is pinkish (Waldron 1984, Arny 2006, eFloras 2008). The twigs are smooth, grey to light brown, and 1.5 to 4.5 mm in diameter (usually 2 to 3.5 mm). Terminal buds are smooth (or with faintly hairy scales), grey to greyish-brown or dark brown, oval (broadest at or above the middle), 4 to 8 mm long (usually 6 mm) and often noticeably five-angled in cross section (Morsink and Pratt 1984, eFloras 2008). The leaf buds are oval, pointed, and typically have some soft, grey hairs (Waldron et al. 1987). The leaves are oblong or oval (with the narrower end at the base), and have three to eleven (usually 7 to 9) opposite lobes with bristle-like acute teeth. The sinuses are variably deep, but are typically cut more than half-way to the midrib (Morsink and Pratt 1984, Waldron 1984, Arny 2006, eFloras 2008). The very wide, deep sinuses are distinctive. They are often bigger than Black Oak sinuses, cut almost to the midrib and sometimes tightly closed at the ends, giving them an oval appearance (Waldron pers. comm. 2013b). Leaves are lustrous dark green above and paler green below with conspicuous tufts of rusty hairs arranged in a radiating pattern in the vein axils on the otherwise smooth underside of the leaf. Autumn leaves of Shumard Oak are noted for their deep crimson or wine red colour, which may have led early naturalists to mistakenly identify it as Scarlet Oak (Quercus coccinea), a species that does not occur in Ontario (Morsink and Pratt 1984, Waldron 1984, Arny 2006, eFloras 2008). Like Hill’s Oak (Quercus ellipsoidalis) and Pin Oak (Quercus palustris), the leaves persist into winter, and may remain green longer than those of other oaks (Waldron et al. 1987). In southern portions of the range, leaves can be up to 20 cm long and 15 cm wide (Arny 2006, eFloras 2008). However, in Ontario leaves are rarely longer than 18 cm except on vigorous vegetative shoots (Waldron pers. comm. 2013b).
Flowers are produced when the leaves emerge in the spring. Male and female flowers are found on the same tree but are produced separately. The staminate (male) flowers occur as slender, drooping, hairless catkins. The pistillate (female) flowers are solitary, or occur in pairs on short stems (Waldron 2003). The smooth, oblong to egg-shaped (sometimes almost round) acorns are 14 to 30 mm long and 10 to 20 mm wide, are large compared to other oaks and require two years to reach maturity (Waldron 1984, Arny 2006, eFloras 2008). Less than one third of the acorn is covered by a flat-topped cap, which has overlapping grey, smooth or slightly hairy tightly-pressed scales. The inner surface of the cap is light-brown to red-brown and smooth, or with a ring of fine hairs around the scar. The scar is 6.5 to 12 mm in diameter.
Shumard Oak reportedly hybridizes with at least 12 other species, of which Pin Oak (Q. palustris; = Q. × mutabilis E. J. Palmer & Steyermark), Red Oak (Q. rubra), and Black Oak (Q. velutina; = Q. × discreta Laughlin) occur naturally in Ontario (eFloras 2008). Shumard Oak is most closely related to Blackjack Oak (Q. marilandica), which does not occur in Ontario (eFloras 2008) and Black Oak (Guttman and Weigt 1989). The ability to identify between Shumard Oak and possible hybrids is essential to the informed management for the species. A table comparing key features of Shumard Oak and related oaks is provided in Appendix 1.
Kartesz (1999) recognizes several varieties of Q. shumardii as distinct, including variety schneckii and variety stenocarpa. The 1997 Flora of North America did not recognize these varieties as distinct (NatureServe 2012). eFloras (2008) describes three varieties based on acorn shape and cap size. Trees with shallow cups covering ¼ of the nut are treated as Q. shumardii var. shumardii. Trees with more deeply rounded cups covering ⅓ of the nut are treated as Q. shumardii var. schneckii (Britton) Sargent. The trees in Missouri and Illinois that Laughlin used to describe Q. shumardii var. stenocarpa Laughlin have ellipsoid acorns with very small (5.5 to 7 mm high × 12.5 to 18 mm wide), shallow cups that cover less than ¼ of the nut. In addition, there are at least four registered cultivar names for Shumard Oak, including three that are patented in the US: Q. shumardii 'ACNRT1' ("MADISON ™"), Q. shumardii 'QSFTC' ("PANACHE ®") and Q. shumardii 'Royal Flush' (Trehane 2010).
Recent genetic research into identifying genomic simple sequence repeat (gSSR) markers and simple sequence markers derived from expressed sequence tags (EST-SSR) for red oak species in North America shows that genetic markers could be used to identify species, but the suite of genetic markers for Shumard Oak has not yet been identified and tested (Sullivan et al. 2012).
Species biology
Oaks are wind-pollinated and require pollen from neighbouring trees to produce seed (Knapp et al. 2000). The density of pollen grains declines rapidly with increasing distance from the source so isolated oak trees may receive insufficient quantities of pollen, reducing acorn production (Knapp et al. 2000). However, oaks typically produce large volumes of pollen so trees may need to be far apart before reproductive ability is severely affected. The optimum density of trees for pollination is not known (Knapp et al. 2000). Knapp et al. (2000) found that the most isolated Blue Oak (Quercus douglasii) trees in their study area (60 m from their nearest neighbour) still produced acorns, but in significantly lower quantities than trees with multiple pollen-producing neighbours. It is likely that isolated Shumard Oak trees, such as those found along fencerows in heavily deforested areas such as Essex County, are producing low quantities of acorns.
Flowering occurs in March and April, sometimes into June. Acorns are produced in September and October (USDA 2009). Shumard Oak does not produce acorns until it is at least 25 years old. Large acorn crops are produced every two to three years, with optimum production at about 50 years of age (Sullivan 1993, Bonner 2008a, USDA 2009). Acorns with multiple embryos are often produced (Edwards 1990). Shumard Oaks are known to have reached at least 480 years of age (USDA 2009). The long generation time makes breeding programs difficult to maintain and they are often abandoned (Aldrich and Cavender-Bares 2011).
Acorns are an important food source for many birds and mammals. In the fall, acorns comprise up to 70% of the diet of White-tailed Deer (Odocoileus virginianus) and Wild Turkeys (Meleagris gallopavo) (Demers et al. 2011). Hard mast (hard-shelled seeds), mainly acorns, makes up 67% of the diet of Blue Jays (Cyanocitta cristata) from October to March and 43% of their overall annual diet. Blue Jays prefer "green" acorns (acorns that are still attached to the tree) but will also forage on the ground after most of the acorns have fallen. These birds actively select acorns that are not infested with weevils (Tarvin and Wooifenden 1999). Several of the woodpecker (Picidae family) species that occur in southern Ontario are also known to eat acorns (Shackelford et al. 2000, Smith et al. 2000, Jackson and Ouellet 2002, Bull and Jackson 2011). Acorns from trees in the red oak group are typically higher in protein, fat, and calories than white oak group acorns but are also typically higher in fibre and tannins, which make them more bitter-tasting and harder to digest. Acorns from the white oak group are only available to terrestrial wildlife for a short period of time because they tend to germinate soon after they fall from the tree. Red oak acorns remain dormant and available to wildlife for a longer period of time and are an important food source in late winter, when white oak acorns have germinated and most other food sources are scarce.
Shumard Oak acorns are exceptionally high in protein (Ober 2011). Songbirds, squirrels (Sciurus and Tamiasciurus spp.) and other rodents, White-tailed Deer and Wild Turkeys have been documented eating Shumard Oak acorns (Sullivan 1993). Blue Jays and grackles (Quiscalus spp.), including the Common Grackle (Quiscalus quiscula), are known to dig up and consume acorns of other oak species (Peer and Bollinger 1997, Bonner 2008a) and likely eat Shumard Oak acorns. All of these wildlife species are abundant throughout the Ontario range of Shumard Oak. Shumard Oak acorns are a preferred food of Wood Ducks (Aix sponsa) (Schaefer et al. 2012).
Several species of common birds and mammals cache and bury acorns for future consumption, which helps with dispersal and germination of Shumard Oak and other oaks. Blue Jays have been documented as moving 54% of pin oak acorns over one kilometre on average (Aldrich et al. 2005a). During a radio telemetry study, each Blue Jay cached 3,000 to 5,000 acorns in one autumn (Tarvin and Wooifenden 1999). Squirrels are also help oak dispersal (Aldrich et al. 2005a). Differences in the foraging and caching behaviour between species likely affect dispersal and genetic structure of oak populations. Grey Squirrels (Sciurus carolinensis) are scatter-hoarders, dispersing a few acorns to each of many sites, , mixing them with seeds from different trees. Red Squirrels (Tamiasciurus hudsonicus) are larder-hoarders, dispersing numerous acorns to each of few sites, keeping seeds from the same tree together (Aldrich et al. 2005). Blue Jays bring several acorns to a cache site but bury each acorn separately within a one to three metre radius (Tarvin and Wooifenden 1999). Larder hoarding is much less effective at fostering oak regeneration than scatter hoarding (Goheen and Swihart 2003 as cited in Aldrich et al. 2005b)
Acorns of the red oak group have variable dormancy caused by the pericarp, the embryo, or both (Bonner and Vozzo 1987). Acorns require cold stratification (winter-like conditions) to germinate. Acorns that are sown in the fall or early winter do not require artificial stratification to stimulate germination while acorns sown in the spring and summer may, depending on how they are stored (Bonner 2008a). Seed dormancy can be broken by stratifying the seeds for 60 to 90 days at one to five degrees Celsius (USDA 2009). Bonner (2008a) describes methods of stratifying acorns and testing germination prior to sowing. To test germination of intact Shumard Oak acorns, the acorns require 60 to 120 days of cold stratification followed by 29 to 50 days of daytime temperatures of 32°C and nighttime temperatures of 21°C. Most other red oaks require less than 90 days of cold stratification followed by daytime temperatures in the 24 to 30°C range and nighttime temperatures between 16 and 20°C. This suggests Shumard Oak requires warmer springtime temperatures to stimulate germination than other red oaks and that it is limited to a more southern distribution. Shumard Oak acorns with moisture content below 20 to 30 percent are unlikely to germinate (Olson 1974).
Most species of the red oak group are temperate-recalcitrant. Acorns can be stored for up to three years by maintaining high moisture levels (above 30%) and storing just above freezing (1 to 3°C) in containers that allow some gas exchange with the surrounding air (Bonner 2008a). Most red oak species acorns will germinate in storage under these conditions, but pre-sprouting does not prevent sowing or production of plantable seedlings (Bonner 1982 as cited in Bonner 2008a). Detailed instructions for storing and germinating acorns are available in Bonner (2008a) and Connor (2004).
Like most oaks, Shumard Oak is able to repeatedly regenerate from the stump or root collar if the main trunk is cut or girdled (Buckley and Evans 2004, Van Lear 2004). This is thought to be an adaptation to fire. First Nations used fire to manage the landscape for thousands of years prior to the arrival of Europeans. The extent of the area burned by First Nations prior to European settlement is disputed (Patterson 2006), but they may have maintained much of eastern North America as open woodlands, prairie, and savannah (Van Leer 2004). Due to the high burning frequency, fires would have been low to moderate intensity surface fires that would have killed the above ground portions of young trees without harming the root (Van Lear 2004). Fire can also reduce the impacts of weevils on acorns, and there is some evidence that First Nations used fire for this purpose (Riccardi et al. 2004). Under such a regime, oaks would be the dominant forest trees (Van Lear 2004), as they tend to have larger root systems than Red Maple (Acer rubrum) and Tulip Tree (Liriodendron tulipifera), two major competitors insouthern forests (Buckley and Evans 2004). However, maple sugar was also an important part of First Nations diet and trade in the northern part of Shumard Oak’s range, so a mix of fire-maintained oak forests and mature maple forests, which do not develop in areas of frequent fire, would likely have been present in southern Ontario. As a lowland species (Waldron et al. 1987, Waldron 2003), Shumard Oak may have been less affected by First Nations fire regimes than upland oak species.
Oak stump sprouts can regenerate oak forests when reproduction from seed is lacking and few oak seedlings and saplings survive into larger size classes due to suppression, competition, deer browsing and other factors. Oak stump sprouts grow faster than oak seedlings and saplings because the extensive residual root system of stumps store large amounts of carbohydrate (energy) reserves. The faster rate of growth helps them become established in the regeneration layer and to grow out of the browse reach of deer faster than saplings and seedlings waiting in the understory (Buckley and Evans 2004). Rapid height growth in the first year of the canopy opening is critical for subsequent survival on sites where deer browsing, frost damage, competition for light and other factors are intense near the ground (Buckley and Evans 2004). Regenerating from the stump enables Shumard Oak to take advantage of the new opening in the canopy (created by the fallen tree) more effectively than seedlings and saplings.
Buckley and Evans (2004) found that the timing of tree cutting and the diameter of the tree affects the vigour of Shumard Oak sprouts that grow from the stumps of young trees. Stumps that were cut between the last week of July and the end of January produced sprouts that grew in height and diameter much faster than stumps cut in March and May, with the most vigorous sprouts produced from stumps cut in September and November. After four months of growth, the mean height of sprouts in the September cutting treatment was nearly twice the mean height of sprouts from the May treatment. Larger diameter stumps tended to produce more vigorous sprouts than smaller stumps, but the difference was not statistically significant. Shumard Oak and other red oaks tend to produce fewer sprouts from cut stumps than the white oaks, but the sprouts tend to be significantly larger.
The variation in sprout vigour with season and stump size during the first year after cutting is likely due to the amount of carbohydrates stored in the roots (Buckley and Evans 2004). Carbohydrate reserves would be at their peak in late summer and early fall and at their lowest in the spring. Larger stumps would have larger energy stores than smaller stumps because their root systems would be more extensive. Wilson (1968) found that overstory competition may also reduce the growth of Red Maple sprouts from stumps cut during the growing season. This is likely true for Shumard Oak and other species as well. Buckley and Evans (2004) concluded that it was preferable to cut Shumard Oak in the fall in order to stimulate regeneration by coppicing (cutting back) or to maximize the growth of sprouts from stems damaged during harvesting.
Oaks (in general) form beneficial associations with micorrhyzal (soil-inhabiting) fungi (Aldrich and Cavender-Bares 2011). The fungi attach to the roots and act like root hairs. It increases the host tree’s water and nutrient absorption, helps tolerate low pH and toxic levels of metals, and provides an additional layer of protection against some pathogens, all of which enable the tree to grow faster than it would without the fungi (Ward 1892, Davis and Jacobs 2004). The fungal associates vary with the oak species. Inoculating the roots of planted oaks with an associated micorrhyzal fungus species has improved growth and survival rates (Davis and Jacobs 2004). However, these fungal associations can also play a role in disease transmission between oaks in the same stand (Canadian Forest Service 2012b, Cook 2012).
2.2 Population and distribution
Shumard Oak is found in the eastern half of North America from New York and Pennsylvania south to Florida, west to eastern Texas, and north to Nebraska, Missouri, Illinois, and Michigan. In Canada, the species is found only in southern Ontario (NatureServe 2012). Although oak enthusiasts had been aware of suspiciously Shumard-like oaks in southwestern Ontario since at least the 1960s (Meyers 1984), and specimens collected in 1979 were tentatively identified as Shumard Oak (Waldron pers. comm. 2013a), it was not officially documented in Ontario until 1983 (Waldron 1983). Table 1 summarizes the current conservation status throughout the species' range.
Table 1. Conservation status of Shumard Oak by state and province.
| S Rank | State/ Province |
|---|---|
| 1 | New York (var. shumardii), West Virginia |
| 2 | Maryland, Michigan, Pennsylvania, |
| 3 | Illinois, Ontario |
| 4 | North Carolina, Virginia (var. shumardii) |
| 5 | Arkansas, Indiana, Kentucky, |
| NR | Alabama, Florida, Georgia, Kansas, Louisiana, Mississippi, Missouri, Nebraska, Ohio, Oklahoma, South Carolina, Tennessee, Texas, |
| U | Alabama (var. shumardii) |
Source: NatureServe 2012
In Ontario, the Shumard Oak is restricted to Essex, Lambton and Elgin counties and the regional municipalities of Chatham-Kent and Niagara (COSEWIC 1999, Oldham 2010). Most of the records are for Essex County (NHIC 2012). In Lambton County, the species is most common on the Lambton clay plains in the south-central part of the county (Mills and Craig 2008). The identity of some of the individuals in Niagara is uncertain. With the exception of the Navy Island National Historic Site population, some or all may actually be Q. palustris X Q. rubra hybrids (P.W. Ball pers. comm. in Oldham 2010, Garofalo pers. comm. 2012). Before the species was discovered on the US side of the Niagara River in 2001 (Eckel 2002), the nearest confirmed populations to the Niagara region were over 200 km away.
Table 2 summarizes the current known Ontario distribution and abundance data. Tree size and abundance were usually, but not always, recorded. Many historic locations consisted of single, relict trees along fencerows and roads or in fields. At the time of the last COSEWIC status report, there were fewer than 40 known locations and the provincial population was estimated to be well below 1,000 trees, and "may not even exceed 250" (COSEWIC 1999). Six new element occurrences (EOs) were identified and confirmed between 1999 and 2004 and another 11 new locations have been documented since 2002. In addition, there are 25 unprocessed recent (1983 – 2011, but most since 2000) observations in the NHIC Central Holding database that have not yet been incorporated into Biotics. Some of these new locations and observations may be new occurrences, others are likely just additional locations within known occurrences. Several new occurrences are of multiple trees and some of these sites have evidence of regeneration.
Table 2. Distribution and abundance of Shumard Oak in Ontario
| Upper Tier Municipality (total # of EOs) (EO Rank) EO ID | Number of EOs (Rank – Year of Last Observation) | Comments |
|---|---|---|
| Regional Municipality of Chatham-Kent (1) (H) 2159 |
1 (H - 1983) | One 1983 record from a conservation area. Conservation authority staff have since found a few old trees in a low lying mucky area in same conservation area. The trees had buttressing at the base and coppice growth on the trunk. Identification was not certain, but habitat and buttressed trunk match Shumard. Date and count of trees was unavailable (Van Wagner pers. comm. 2013). May also occur in Rondeau Provincial Park. Woodliffe (pers. comm. 2013) reported seeing trees that "look suspicious," but could not confirm the identity. |
| Elgin County (1) (E) 21125 | 1 (E - 1993) 1 (new - 2005) |
The only record for the existing EO near West Lorne (1993) lacks abundance information, but is likely for a single mature tree; acorns were collected. No abundance information is available for a possible new record found near Dutton in 2005. |
| Essex County (22) (H) 2151, (H) 2153, (H) 2154, (H) 2155, (E) 2156, (H) 5401, (H) 5402, (H) 5403, (H) 5404, (H) 5405, (H) 5406, (H) 21113, (H) 21114, (H) 21115, (H) 21116, (H) 21117, (E) 21126, (H) 21127, (H) 21129, (CD) 21130, (E) 64430, (E) 66321 |
2 (H - 1979) 1 (H - 1980) 6 (H - 1982) 3 (H - 1983) 4 (H - 1985) 1 (H - pre 1984) 1 (CD - pre 1984) 3 (E - 1993) 1 (E - 2001) 1 (new - 1993) 1 (new - 2005) |
Essex County was heavily deforested before Shumard Oak was officially documented in 1983. By then, less than 2% of the original forest cover remained. There was probably a large population of Shumard Oak in the county before deforestation (Morsink and Pratt 1984). Most historic records are for isolated mature trees on roadsides or in relict woodlots and hedgerows on private property. Sites have probably not been searched since the last observation date. Three extant EOs are in protected areas. One EO is an isolated tree in a private wildlife sanctuary. A Windsor city park has "some large mature specimens." There are many impressive mature trees at Maidstone Conservation Area (Waldron pers. comm. 2013b). A potential new record of about 50 large trees was found in a remnant woodlot on private hunt club property in 2005. Most trees were 30 to 40 cm diameter at breast height (dbh), with two approaching 71 cm dbh. The landowners are in the process of reforesting the former agricultural land on the property. The "new" 2005 record is probably part of the extant population that was observed in 1993. |
| Lambton County (9) (H) 2162, (H) 5407, (H) 21112, (H) 21123, (H) 21124, (E) 32484, (E) 32485, (E) 32486, (E) 32487 |
4 (H - 1985) 1 (H - 1987) 2 (E - 2000) 2 (E - 2001) 1 (new - 2003) 1 (new - 2005) 4 (new - 2007) |
All historic EOs are on private property. Two EOs were single, isolated roadside trees. These sites are not believed to have been searched since the 1980s. The extant EOs are for groups of trees and all have evidence of natural regeneration. At three sites, tree sizes ranged from polewood to trees over 60 cm dbh. The fourth site had saplings to mature trees over 50 cm dbh. Red Oak hybrids were observed at one site. The species was found at Bickford Oak Woods Conservation Reserve in 2003. Several trees were found on the Bowens Creek Lands, which the St. Clair Region Conservation Authority (SCRCA) manages for the county. Two of the potential new sites found in 2007 are private property. A very large tree (about 1 m dbh) was found at one site. The other site has a 40 to 45 cm dbh tree on the edge of a woodlot and several trees in a nearby hedgerow surrounded by cropland. A single large tree (50 to 60 cm dbh) was found in the McKeough Dam Conservation Area and several trees in all size classes, including some trees over 62 cm dbh, were found at the nearby Reid Conservation Area in 2007. A small number of Shumard Oak occur on Walpole Island (Woodliffe pers. comm. 2013). |
| Regional Municipality of Niagara (5) (H) 2157, (E) 2158, (H) 2160, (H) 2161, (E) 92636 |
2 (H - 1980) 1 (H - 1983) 1 (E - 2003) 1 (E - 2004) 1 (new - 2003) 1 (new - 2007) |
The identity of some of the historic EOs near Niagara-on-the-Lake is disputed. One of the extant EOs is on Navy Island National Historic Site. The other extant EO is on private property. Single, isolated trees were found at three different locations on a former Department of National Defence (DND) property in 2003. This property was transferred to Parks Canada in the 1940s but management is shared between Parks Canada and DND due to unexploded ordnance. These three locations likely represent a single new EO. Another potential new EO was discovered by MNR staff in 2007. |
Figure 1. Historical and current distribution of Shumard Oak in Ontario
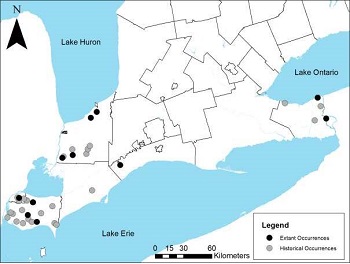
Enlarge figure 1. Historical and current distribution of Shumard Oak in Ontario
The five municipalities have been searched by several individuals at various times (COSEWIC 1999), and natural heritage inventories have been conducted for many significant environmental areas. Thorough inventories for Shumard Oak have not been conducted. Most of the woodlots with potential habitat are privately owned. About the time that Shumard Oak was confirmed in Ontario, the Ministry of Natural Resources began significant reductions to the private land forestry program in southern Ontario and eventually ended the program (Woodliffe pers. comm. 2013). Consequently, there were fewer people on the landscape to notice the species, and most of these private woodlots have not been searched.
Most of the Areas of Natural and Scientific Interest (ANSI) in southwestern Ontario that have lowland forest may have Shumard Oak, and the species is probably more abundant and widespread than indicated by the available records (Woodliffe pers. comm. 2013).
2.3 Habitat requirements
Like all oaks, Shumard Oak requires the full sunlight of open habitat conditions for seedling establishment (COSEWIC 1999). Shumard Oak grows on moist, well-drained loamy soils of stream and river terraces, on adjacent ridges and bluffs, and on mesic (moist) slopes and poorly drained uplands at elevations from 0 to 500 m (eFloras 2008, USDA 2009). Even though it is found on mesic sites, it is intolerant or only weakly tolerant of flooding and does not usually occur on the lowest river bottoms (Sullivan 1993, USDA 2009). In Ontario, it has been found on mesic-hydric (moist to wet) sites with clays and clay loams of the Brookston, Perth, Toledo and Lincoln soil series (Waldron et al. 1987). These soil types and their distribution in Canada are described in Soil Classification Working Group (1998) and on the University of Saskatchewan’s Soils of Canada website (http://www.soilsofcanada.ca/index.php). These same poorly-drained soil types are found in the Michigan, Indiana and Ohio portions of the Shumard Oak range. Soils in the Ontario portion of the range are rich in all nutrients except phosphorus (Waldron et al. 1987). Shumard Oak is found on slightly elevated spots within these low, wet areas (Waldron 2003).
While the species naturally occurs only on wet sites, it also grows well when planted on much drier clays and loams. It will grow on both acidic and slightly alkaline soils. Shumard Oak is tolerant of air pollution, salt, soil compaction and drought (Sullivan 1993, Waldron 2003). Its tolerance and adaptability, in conjunction with its sturdiness, attractive form and fall colour, and its potential for fast growth, make it desirable for restoration programs and as a street tree (Waldron 2003). In Ontario, Shumard Oak has been found in deciduous woods on clay soil, open clay floodplain woods, Sugar Maple forest on silty loam, oak – Sugar Maple forest with swampy areas, mature White Oak – Black Cherry – Ironwood – Red Oak forest on clay, Red Oak – Red Maple – White Ash – White Oak forest and Swamp White Oak mineral deciduous swamp (NHIC 2012). However, consistent, standardized habitat descriptions are lacking for Ontario populations.
Throughout its range, Shumard Oak is usually widely spaced and never occurs in pure stands. Common tree associates in the United States include White Ash (Fraxinus americana), Shagbark Hickory (Carya ovata), Shellbark Hickory (C. laciniosa), Mockernut Hickory (C. tomentosa), Bitternut Hickory (C. cordiformis), Water Hickory (C. aquatica), Blackgum (Nyssa sylvatica), Winged Elm (Ulmus alata), Magnolia (Magnolia grandiflora), Tulip Tree (Liriodendron tulipifera), American Beech (Fagus grandiflora), Spruce Pine (Pinus glabra) and other oaks (Sullivan 1993). Common tree associates in Ontario include Shellbark Hickory, Bitternut Hickory, Red Ash (Fraxinus pennsylvanica), Pumpkin Ash (F. profunda), Silver Maple (Acer sacchariunum), White Elm (Ulmus americana), Swamp White Oak (Q. bicolor), Burr Oak (Q. macrocarpa), Chinquapin Oak (Q. muhlenbergii) and Pin Oak (Waldron 2003).
2.4 Characteristics contributing to vulnerability of species
As noted above, acorns are an important food source for many birds and mammals. A large proportion of acorns (up to 90% of the normal crop) are destroyed by weevils (Riccardi et al. 2004). As a result, oak recruitment is probably naturally relatively low. The high protein content of Shumard Oak acorns may make its acorns particularly attractive to acorn-eating wildlife species that are abundant in southern Ontario.
All oak acorns are recalcitrant seeds. This means they will not germinate if they dry out. They require a relatively high minimum moisture content and are thus difficult to store.
Oaks in general are host to numerous herbivorous and gall forming insects (Edwards 1990, Aldrich and Cavender-Bares 2011), including several species of leaf-tying caterpillars and several species of leaf chewing insects (Marquis and Lill 2010). In addition, at least 42 native borers (Agrilus genus) throughout the United States use oaks as larval hosts (Nelson et al. 2008 as cited in Haack et al. 2009). At least nine occur in Michigan (Haack et al. 2009) and probably also in Ontario.
The Two-lined Chestnut Borer, A. bilineatus, is the most lethal of the native Agrilus species that attack oaks in eastern North America, particularly when the trees are stressed, and is a contributing factor to oak decline in North America (Ciesla 2003, Haack and Benjamin 1982, Haack et al. 2009). It is one of the first borers to attack weakened trees; other species usually attack the tree a year or two after the initial A. bilineatus infestation. A. bilineatus larval galleries girdle weakened trees, killing them within three years of the initial infestation. However, A. bilineatus rarely attacks vigorous trees. When it does, the tree is able to defend itself and the larvae do not survive long enough to harm the tree (Haack et al. 1982, Haack and Benjamin 1983).
The young bark is relatively thin and easily damaged by mechanical impacts and fire (Gilman and Watson 1994, USDA 2009). Basal wounding by fire usually kills at least the top of young trees, either directly by girdling the tree or indirectly by subsequent infection by wood-rotting fungi. However, Shumard Oaks that are only top-killed will regenerate root sprouts (Sullivan 1993, USDA 2009).
Oaks (in general) form beneficial associations with micorrhyzal (soil-inhabiting) fungi (Aldrich and Cavender-Bares 2011) and could be affected by introduced earthworms, which alter the chemistry and micorrhyzal composition of soils (Hendrix and Bohlen 2002).
3.0 Threats
Natural ecosystems are continually evolving in response to a variety of forces and factors. But they are limited in their ability to adapt to rapid change, such as that introduced through human activities. Humans sometimes disrupt and degrade biodiversity through habitat loss, introduction of invasive species, population growth, pollution, unsustainable use and climate change. Our growing population combined with our rising levels of resource consumption can threaten biodiversity (OBC, 2011). Recently, an assessment of pressures on Ontario’s biodiversity showed that many threats are increasing (OBC, 2010b).
The following threats have been identified as currently facing the Shumard Oak in Ontario:
Habitat loss, habitat degradation, and loss of individuals/populations
Shumard Oak is found in the most densely populated region of Ontario. As of 2006, only 3,050 km2 of forest, 222 km2 of wetland, 40 sq. km2 of shrubland, and 12 km2 of grassland remained in the 23,805 sq. kilometre Lake Erie Lowland ecoregion, the ecoregion with all of the Ontario Shumard Oak records. The rest of the land base in the ecoregion is primarily cropland (Filoso and Larocque 2010). The four counties where Shumard Oak occurs have had among the highest rates of forest clearing in Ontario. Less than 3% of the land cover in Essex County, where the species appears to be most common, was forest in the 1970's (Waldron 1984). Over 98% of the historically forested area was converted to other land uses (Morsink and Pratt 1984). Forest cover has been increasing since then (e.g., up to 5% of the land base in 2002), thanks to restoration and conservation efforts by local agencies (ERCA 2013). However, although Essex Region Conservation Authority (ERCA) plants a mix of species at each site (Davies, pers. com. 2012), it would take decades or longer for these plantings to become functioning forest systems depending on the previous land use and their proximity to remnant forests (Woodliffe pers. com. 2013b).
Significant Woodlands and Provincially Significant Wetlands receive some protection under the Ontario Planning Act and the Provincial Policy Statement. However, many woodlots and wetlands have never been evaluated and therefore are not protected from development, even though they might meet the criteria. Significant Woodlands are only protected from activities that require an application under the Planning Act. Activities such as converting a Significant Woodlot to cropland are not restricted by the designation. Municipal tree-cutting by-laws provide some protection from these types of activities, but Chatham-Kent and Essex do not have such by-laws (Ontario Woodlot Association 2003), and enforcement is not always consistent.
The construction of the new Detroit River International Crossing (DRIC) and the widening of Huron Church Road in Windsor will result in the loss or degradation of wet woods along the route (Woodliffe pers. comm. 2013a), with 24 mature Shumard Oak trees potentially being disturbed but no trees actually being displaced (Canada – United States - Ontario - Michigan Border Transportation Partnership 2008).
The remaining woodlots in the Ontario range of Shumard Oak are usually highly disturbed. Disturbance includes summer browsing by cattle and horses, periodic logging, clearing for recreational and home sites, and alterations to hydrology caused by tile drainage, ditching and channelization of watercourses (Waldron et al. 1987). These activities still take place in Essex County and other parts of the Ontario range (Donley pers. obs.). Many of these may be threats to individual trees but, with the exception of hydrological changes, generally are not negative factors in the survival of the species because oaks reproduce well on open, disturbed sites, but not under closed canopies (Waldron et al. 1987).
Ditching and channelization generally have greater impact on surface water than on ground water (Waldron et al. 1987). Ditches and drains lower the water table, making the surface drier (Zammit pers. comm. 2013). Waldron et al. (1987) speculated that these methods of draining agricultural land would not threaten existing trees (provided the trees are not cut down) but would increase the number of hybrids with species associated with drier (e.g., Red Oak, Black Oak) and wetter (e.g., Pin Oak) sites. However, the impact depends on the type of wetland and the size of the drain; a large ditch can quickly drain a marsh. Perched wetlands (wetlands found above the water table on soil that restricts water drainage) that are partially maintained by surface water would be negatively affected by any reduction in surface water (Zammit pers. comm. 2013). Waldron et al. (1987) did not speculate on the impacts to regeneration; the drier conditions would likely prevent seedling establishment.
Installation of tile drains would have a much greater impact on Shumard Oak because they alter groundwater hydrology (Woodliffe pers. comm. 2013a, Zammit pers. comm. 2013). Tile drainage quickly removes the gravitational water to prevent drowning of crops, leaving only capillary water, which is the type of groundwater used by plants. After a storm, the water table in agricultural fields with tile drainage falls to the drainage depth in three to four days; in un-tiled fields it may take several weeks of no rain to drop to that level (Fraser and Fleming 2001). Tile drainage is being installed at a massive scale, especially in Chatham-Kent. There are more applications under the Drainage Act for tiling in Chatham-Kent than in the rest of the province combined. The installation of tile drainage may not directly kill existing Shumard Oak but the change in hydrology would increase the stress on swamp forest species, making them vulnerable to disease and pests and would reduce or prevent regeneration (Woodliffe pers. comm. 2013a). Presence of Shumard Oak and other wet-habitat species at risk should be taken into consideration when reviewing applications under the Drainage Act.
Oaks are more tolerant of fire but less tolerant of shade than maples (Acer spp.) and other hardwoods with which oaks are commonly associated (Sullivan 1993, Aldrich et al. 2005b). Fire suppression, and the subsequent growth of maples and other shade tolerant species, has been identified as one of the causes of oak recruitment failure (Aldrich et al. 2005b).
Loss of individuals: crop land expansion/hedge row removal
Many Ontario Shumard Oaks are found in fencerows and remnant woodlots on farms. Recent high crop prices have stimulated many landowners to increase the size of the cultivated area on their properties by removing woodlots and hedgerows. Hedgerows are also being removed to accommodate large modern farm equipment (Gartshore pers. comm. 2013, Waldron pers. comm. 2013). Such practices have been occurring at an increased rate in recent years due to high commodity prices and the amalgamation of farms, particularly in municipalities that do not have a conservation by-law. Numerous woodlots, fencerows, hedgerows, and rows of trees along drains in Chatham-Kent were razed in the winter of 2012/13 in anticipation of a proposed conservation by-law (Donley, Jalava pers. obs.).
Exotic earthworms
Sutherland et al. (2011, as cited in Sackett et al. 2012) stated that "non-native earthworms are recognized as an emerging threat to temperate North American forest ecosystems, and are considered one of the most globally important agents of change to biodiversity and associated ecological and evolutionary processes." Of the 19 species of earthworms found Ontario, 17 were introduced from Europe and two (Sparganophilus eiseni and Bimastos parvus) were introduced from southern US. None are native to Ontario. Another 20 or more species from Asia, Africa, and South America have been introduced to North America but have not yet been confirmed in Ontario (Evers et al. 2012). Earthworms native to the Great Lakes region, including Ontario, are thought to have been extirpated during the last glaciation period (Tiunov et al. 2006).
Exotic earthworms were probably introduced accidentally during and after European settlement through soil used for ship ballast, in soil with imported plant materials (Tiunov et al. 2006), or as cocoons attached to the roots of imported woody plants (Schwert 1977). Current sources of accidental introductions include the release of worms used for fishing bait, worms that escape from vermicomposters (Tiunov et al. 2006), and in the roots of orchids and other imported plants (Schwert 1977). European settlers may have also deliberately introduced earthworms to "improve" the soil (Evers et al. 2012). Once established, exotic earthworms can be inadvertently transported to new locations by the movement of fill for construction activities or compost and other gardening materials, in mud on tires of equipment and off-road vehicles (Evers et al. 2012, Tiunov et al. 2006), and, most commonly, through dumped fishing bait (Cameron et al. 2007, Evers et al. 2012). Based on the range maps in Evers et al. (2012), 15 of the introduced species are found throughout the Ontario range of Shumard Oak.
Ontario forest species are adapted to the soil structure typical of worm-free environments: thick organic horizons (litter layer and duff layers) on top of mineral soil with low organic content. Earthworms completely alter this structure by consuming the duff and by thoroughly mixing the organic and mineral soil layers (Frelich et al. 2006, Tiunov et al. 2006, Evers et al. 2012). Deep burrowing species such as the Night Crawler (Lumbricus terrestis, also commonly known as the Dew Worm) can completely break down the thick duff layer and incorporate it into the soil within just a few years (Sackett et al. 2012). Earthworms also:
- consume the organic content of the mineral soil layers (Sackett et al. 2012);
- make the soil denser by incorporating the thick duff layers into the mineral soil, reducing the thickness of the forest floor and cementing soil particles together (Frelich et al. 2006, Evers et al. 2012), negatively affecting plant roots;
- reduce soil nitrite and nitrate levels and phosphorus levels in the soil layers with the highest concentration of fine roots (Frelich et al. 2006, Evers et al. 2012);
- alter soil micro-organism communities and soil nutrient cycling by mixing the soil layers and predating microbial species (Frelich et al. 2006, Evers et al. 2012);
- alter soil hydrology with their burrows, which greatly increases the water infiltration rate, and by eliminating the duff layer, which normally acts as a buffer against summer drought by absorbing rain water and slowly releasing it into the soil (Evers et al. 2012); and
- disrupt root systems and mycorrhizal relations with their burrowing activities, reducing seedling establishment (Evers et al. 2012).
Exotic earthworms have reduced the duff layer in Ontario forests to the point where trees have no buffer against summer drought and no insulation against winter freezing. These conditions stress the trees, making them more vulnerable to diseases and pests (Gartshore pers. comm. 2013). Regeneration of acorns is reduced because they dry out and freeze and the lack of a duff layer causes increased seed predation (Evers et al. 2012, Gartshore pers. comm. 2013). Earthworms also change the fungal processes considerably (Hendrix and Bohlen 2002, Evers et al. 2012, Woodliffe pers. comm. 2013a), often resulting in the extirpation of strongly mychorrhizal herbaceous plant species (Frelich et al. 2006). Since Shumard Oak, like all oaks, benefits from a symbiotic relationship with mychorrhizal fungi, it would be particularly vulnerable to such changes. The increased soil density and water infiltration rates caused by earthworm activities combined with the elimination of the duff layer could result in more xeric (drier) site conditions, which would negatively affect survival and regeneration of mesic species like Shumard Oak (Frelich et al. 2006, Evers et al. 2012).
Other invasive species
The European Gypsy Moth (Lymantria dispar) was deliberately introduced to the eastern US near Boston in 1869 as a potential source of silk. It subsequently escaped and gradually spread, becoming established in Ontario in the late 1960s (Nealis and Erb 1993). The spread was slow because of decades of quarantine and eradication measures in the northeastern US and because the females are flightless, restricting the species to a naturally slow rate of spread (Nealis and Erb 1993, Krcmar-Nozic et al. 2000). The gypsy moth is a defoliator that prefers all types of oak, including Shumard Oak, but will attack many other hardwood species as the number of caterpillars increases (Krcmar-Nozic et al. 2000, Canadian Forest Service 2012b).
European Gypsy Moth females lay spongy egg masses covered with tan or buff- coloured hairs on the trunks and branches of trees. The species overwinters in the egg stage. In spring, newly hatched larvae feed by chewing small holes in the surface of the leaves, while older larvae eat the entire leaf. Mature larvae climb down the tree and hide in shady places, usually in the leaf litter at the base of the host tree, during the day, and climb back up the tree to feed at night. When the tree is completely defoliated, the larvae leave the tree and migrate in groups in search of a new host. During severe outbreaks, trees and shrubs are completely defoliated over large areas. Understory shrubs and plants may also be attacked. Although trees are able to produce a new crop of leaves over the summer, the damage significantly reduces the trees' growth (Canadian Forest Service 2012b). Between 153,000 and 347,500 hectares were severely defoliated (30% or more of the tree’s current foliage removed) in Ontario during each of the severe outbreak years of 1985, 1986, 1991, and 2002. During other years (1981 to 2009), between 5 and 82,000 hectares (usually 13,000 or less) were defoliated annually in Ontario (Canadian Forest Service 2012a).
The Rosy Gypsy Moth, Lymantria mathura, is an Asian species that has become established in North America. Unlike the European Gypsy Moth, the females are not flightless, which has enabled the species to spread much faster than its European counterpart. This species also targets oaks (Canadian Forest Service 2011a).
Non-native cool-season grasses are often used on roadsides and lawns and may be impacting oaks in hedgerows, field edges, residential areas, outdoor sports complexes, and urban parks. Some species are allelopathic and prevent regeneration. Other species directly impact trees by enveloping roots or spearing tap roots. Indirect impacts include drying out surface soils, harbouring pests and rodents and competition for nutrients (Gartshore pers. comm. 2013). These stresses may make oaks more susceptible to disease and pests and negatively affect natural regeneration.
European Oak Borer (Agrilus sulcicollis) (EOB) is a non-native pest of oak species. It is widespread in Europe, where it primarily infests oaks but has occasionally been found on other deciduous trees (Haack 2010). This species was first identified in North America in 2008 from a specimen collected on a Red Oak at Pinery Provincial Park (Lambton) in 2006 (Jendek and Grebennikov 2009). The species was then found in New York and Michigan during the 2009 US national Emerald Ash Borer (Agrilus planipennis) (EAB) survey. Unidentified specimens in various museum and private insect collections were subsequently reviewed and the species was found to have been collected in Essex, Elgin, Middlesex, Waterloo and Lambton Counties between 1995 and 2008, and in Michigan in 2003 (Haack et al. 2009, Jendek and Grebennikov 2009). Most of the collections were from EAB traps on ash trees, but some were captured on oaks (Haack et al. 2009, Jendek and Grebennikov 2009). Targeted searches for EOB were conducted in Ontario from 2009 to 2011. The species is now believed to occur throughout the range of oak hosts in Ontario (Cleland pers. comm. 2013).
Zablotny (2009) provides detailed instructions for distinguishing EOB from a similar looking, widespread non-native Agrilus species that exclusively uses honeysuckle (Lonicera spp.) as a larval host. However, the adults of many Agrilus species are difficult to identify to species. Larval borers can only be identified to genus and must be reared to adulthood to determine species. In addition, it is not unusual to find more than one Agrilus species infesting a single oak tree, and these species are notoriously difficult to identify. For these reasons, suspected specimens must be sent to a taxonomic specialist to confirm identification (Hoebeke 2010).
Little is known about the life cycle of EOB in North America, most of the available information is from Europe. In Europe, adults feed on the leaves and wood of host plants from May to July, peaking between mid-May and late June (Jendek and Grebennikov 2009). Eggs are deposited in bark cracks along the trunk and major branches or on recently cut logs, usually in locations that are exposed to the sun. The larvae tunnel at the interface between the sapwood and the inner bark and, if the bark is thick enough, within the bark itself (Haack et al. 2009, Jendek and Grebennikov 2009). The onset of adult emergence in Europe is the same as EAB in North America (May), but adult EOB flight (in Europe) continues into July whereas EAB flight (in North America) peaks in mid-June. EOB overwinters in the host tree as a full-grown larva.
The adults emerge from the trunk and larger branches through the D-shaped bark holes typical of most Agrilus species and are capable of flying long distances to find suitable hosts (Jendek and Grebennikov 2009, Hoebeke 2010). The species has been collected from trunks of Red Oak in Ontario and from English Oak (Q. robur) logs in Michigan, but the hosts and range in North America have not been fully documented (Haack et al. 2009, Hoebeke 2010).
To date, EOB has not been documented on Shumard Oak. However, since both Agrilus and Quercus species are difficult to identify, and EOB was only recently identified in Ontario, it is possible that the species has been overlooked on Shumard Oak. In Europe, EOB is commonly associated with oak mortality, especially in forests stressed by drought. However, EOB is usually a secondary pest, only attacking trees that are already stressed by drought, defoliation, disease or infestation by other borers (Haack et al. 2009). The species appears to behave the same way in Ontario (Cleland pers. comm. 2013). Since there are many native Agrilus species that use oaks as hosts, Haack et al. (2009) hypothesized that the impact of EOB will be limited by direct competition with other species and by the defenses that North American oaks have developed against Agrilus borers. Consequently, EOB is expected to be far less lethal to oaks than EAB is to ash (Hoebeke 2010). The fact that the species does not appear to be causing elevated levels of oak mortality in Pinery Provincial Park (MacKenzie pers. comm. 2013), where the species has been established since at least 1995, supports this hypothesis.
EAB has caused changes to forest structure and landowner attitudes toward woodlots. Both factors are likely having impacts on Shumard Oak, but such impacts have not been investigated. EAB has opened up the canopy of affected forests, creating light conditions that could promote oak regeneration. However, it is unknown whether there are young Shumard Oaks in the understory that could become established in the canopy before the maples and other shade tolerant species. In addition, EAB, Asian Long-horned Beetle (Anoplophora glabripennis) and other exotic forest pests have changed landowner attitudes and activities in woodlots. Landowners may be more likely to allow timber harvest or firewood operations (Woodliffe pers. comm. 2013). For example, when the first EAB quarantine was established, logging companies implemented advertising campaigns in rural areas of Middlesex County, encouraging landowners to sell the timber in their woodlots while they could. Many woodlots in the county were logged but not completely cleared (Donley, pers. obs.). Landowners with woodlots affected by EAB may decide that, since much of the woodlot is already dead, it is time to convert the land to agriculture (Woodliffe pers. comm. 2013a). This may be contributing to the high rate of woodlot clearing that is currently occurring in areas like Chatham-Kent. The impacts of the ecological and landowner attitude changes caused by EAB and other exotic pests on Shumard Oak needs study.
Winter Moth (Operophtera brumata) is a European species that became established in the Maritime provinces by the 1950s and on Vancouver Island, British Columbia, before 1977. It is a defoliator that attacks several hardwood species, particularly oaks and apples. The yellowish-green larvae hatch on spring mornings and use a thread to ride the rising warm air into the canopy. Newly emerged larvae attack buds, while older larvae consume the leaves, mostly during the last two weeks of June. When the host tree is severely defoliated, the larvae drop to the ground by the thousands and seek out new hosts. If the trees are partially defoliated over four consecutive years, the mortality of infested branches and twigs can be high. Trees may die if complete defoliation occurs over several consecutive years. In Nova Scotia, about 40% of red oak stands have been killed in some areas. Nova Scotia has effectively used two non-indigenous parasites to control Winter Moth. Bacillus thuringiensis, an organic insecticide also used to control Gypsy Moth and other defoliators, is also effective (Canadian Forest Service 2011b). The Canadian Forest Service (2011c) lists Winter Moth as a pest of Shumard Oak. Although Winter Moth has not become established in Ontario (Canadian Forest Service 2011b), the high mortality rate for infested red oak stands in Nova Scotia shows that this species could become a serious threat to Shumard Oak if introduced to Ontario.
The Granulate Ambrosia Beetle (Xylosandrus crassiusculus) (GAB), also known as the Asian Ambrosia Beetle, is known to attack Shumard Oak and over 200 other tree species in North America (Frank and Bambara 2009, LaBonte 2010). The beetle attacked and killed potted Shumard Oak saplings (trees less than 3 meters tall) in a nursery in Florida. The attacked saplings had no other symptoms of disease, insect infestation, or visible stress. The beetles bored into green, fresh, unstained portions of the stems of the Shumard Oak saplings (Atkinson et al. 2005).
Found in South Carolina in the early 1970s, GAB has since spread along the Atlantic and Gulf coasts south to Florida, west to eastern Texas, and north to Maryland (Frank and Bambara 2009). Females bore into twigs, small branches, and trunks of young, thin barked, deciduous trees of susceptible species (Frank and Bambara 2009). Infestations usually occur near the ground on saplings and at bark wounds on larger trees. The beetle attacks apparently healthy trees as well as stressed trees and freshly cut host material. All species of ambrosia beetles excavate a system of tunnels in the heartwood or pith of the host plant, where they introduce and cultivate the symbiotic ambrosial fungus and produce a brood. Ambrosia beetles feed on the fungus, not the wood of the host. The female GAB packs the entrance tunnel with frass (boring dust) from the galleries, slowly pushing out a toothpick-like strand of frass as she excavates. Once the entrance holes are packed with frass, the beetles are protected from parasites and insecticides, making it hard to control infestations (Atkinson et al. 2005, Frank and Bambara 2009). Fungicides are ineffective against the ambrosia fungus. Infested trees often die from the boring damage, the ambrosia fungus blocking the xylem vessels and interfering with vascular transport, or from infection by a secondary fungal pathogen introduced by the beetle. However, healthy, otherwise stress-free trees may survive and outgrow the attacks (Frank and Bambara 2009). GAB is able to survive long distance transportation in cut logs (LaBonte 2010). The species has been intercepted on shipments to Ontario, but has yet to become established in the province. It could easily be introduced through wood pallets, raw lumber, and infested nursery stock.
Other exotic wood-boring insects could pose a significant threat to Shumard Oak if they become established in Ontario. The Oak Splendour Beetle (A. biguttatus, also known as the Two-spotted Wood Borer), native to Asia and widespread in Europe, is contributing to the decline of oaks throughout Europe. This species as rated as "high risk" for potential establishment and spread in North America (because suitable climatic and host materials occur at many North American ports of entry) and are of special concern to the United States Department of Agriculture (USDA) (Ciesla 2003, USDA-APHIS 2011). The USDA also considers Tetropium castaneum, currently found throughout Asia and Europe, and Platypus quercivorus, currently found in Papua New Guinea and parts of Asia, as "high risk" for establishment and spread in North America. Both species are known to infest oaks within their current range (USDA-APHIS 2011). Jackson et al. (2011) provide detailed instructions for detecting these and other species considered high risk for establishment in the US. These measures could likely be applied in Ontario as well.
Subsidized/hyper-abundant herbivores and seed predators
The populations of many seed predators and herbivores are being maintained at unnaturally high levels by the abundant food supplies created by agriculture, human waste, and forestry practices (Waller 2008). Subsidized seed predators (e.g., White- tailed Deer, Blue Jays, squirrels, other rodents, skunks) are preventing regeneration by consuming most or all of the acorns (Marquis et al. 1976, Aldrich et al. 2005b, Riccardi et al. 2004, Gartshore pers. comm. 2013). There is some evidence that, in the absence fire, populations of the weevils that infest acorns (Curculio and Conotrachelus spp.) are also higher than historic levels; the lack of fire may be contributing to the very high rates of weevil-caused acorn destruction that have been reported (Riccardi et al. 2004).
The seedlings produced by acorns that survived the seed predators are threatened by subsidized or overabundant herbivores, particularly White-tailed Deer. Deer browse on understory vegetation, including small trees and seedlings. When deer are overabundant, deer browse can lead to declines in understory tree regeneration (Tanentzap et al. 2010). Throughout their range, White-tailed Deer are dramatically reducing the regeneration of many tree and shrub species, including the red oaks (Marquis et al. 1976, OMNR 1991, Waller 2008). Deer browse may also indirectly reduce oak regeneration encouraging the establishment grasses and ferns that can interfere with tree seedling recruitment (Waller 2008). In a survey of Wisconsin foresters on natural oak regeneration, 80% of respondents identified deer as "a strong to very strong contributor to (oak) regeneration failures" (Wisconsin Council on Forestry 2006a). Assessments of Wisconsin mixed hardwood-conifer plantations found that Red Oak was one of the three species that were most frequently observed to have deer browse damage (Wisconsin Council on Forestry 2006b). The Wisconsin Council on Forestry has identified deer browse as the most significant barrier to successful forest regeneration (Wisconsin Council on Forestry 2013).
On Navy Island, where Shumard Oak is one of the dominant trees, the extremely high deer population (over 300 deer prior to 2011 herd reduction) has browsed all vegetation within their reach, resulting in park-like conditions in some parts of the island: mature trees with short ground cover, few shrubs, and little tree generation (Anonymous 2004, Zoetewey pers. comm. 2013). The island’s winter deer population is still very high (70 to 100 deer, approximately 60 deer per km2, vs. normal density of 6 to 8 per km2) (Allen; Zoetewey pers. comm. 2013).
Incompatible land use/targeted removal of oaks
Acorns, leaves, and buds of some oaks can be toxic to some domestic livestock. Cattle, goats and sheep may experience fatal poisoning if they ingest too many acorns or too much oak browse (Ober 2011). Although livestock pasturing is not as widespread and is far less common than historically, it still occurs to some degree throughout the Ontario range of Shumard Oak (Donley, pers. obs.). Consequently, oaks may be thinned or completely removed from livestock pastures, including woodlots used for pasture. Since oaks are difficult to identify, Shumard Oak could be mistaken for other, more common red oak species and inadvertently cut down. The practice of removing oaks from livestock pastures was probably more common historically and may have contributed to the historical decline of the species.
Motorized off-road vehicles
Unauthorized All-Terrain Vehicle (ATV) trails are causing erosion in one of the Vegetation Management Units occupied by Shumard Oak (Saint Clair Region Conservation Authority (SCRCA) 2010). While this may not affect mature trees, it could prevent regeneration by destroying seedlings. ATVs also spread invasive species, the seeds of which become embedded in mud that is stuck to the tires and are transported to other sites (SCRCA 2010).
Unsustainable use
Native North Americans had many uses for the bark and acorns of oaks, probably including Shumard Oak (Krochmal and Krochmal 1982). Although no specific reference to such activities was made by the representatives of the First Nations that were contacted for this study (Porchuk pers. comm. 2013), collecting acorns for human consumption and use by enthusiasts of traditional food, crafts and natural products is likely occurring within the Ontario range of Shumard Oak.
Since Shumard Oak occurs in the most densely populated area of Ontario, is often found along fencerows and roads where it is easily accessible. It can be difficult to distinguish Shumard Oak from other oaks in the red oak group and the average person is not versed in oak identification or even aware of the species. Because of this, Shumard Oak acorns could be inadvertently collected. Since there are estimated to be fewer than one thousand Shumard Oak in Ontario, collection of Shumard Oak acorns for any purpose other than cultivation may threaten the ability of the species to maintain and expand its populations.
Wood from oaks in the red oak group, including Shumard Oak, is usually not separated by species at mills or lumber yards. Instead, it is mixed together and marketed as "red oak". It is used for veneer, cabinets, furniture, flooring, interior trim, posts, cross ties, and lumber (Sullivan 1993, NHIC 2012). Since Shumard Oak can be difficult to distinguish from other red oaks, individuals could be inadvertently cut down (COSEWIC 1999). Shumard Oak wood is strong, hard, heavy, and close-grained and is considered superior to the wood of other red oak species (Sullivan 1993). Consequently, Shumard Oak could be targeted by knowledgeable buyers of standing timber.
Disease
Oak reproductive failure has been widely reported and is a concern in many places. Causes include drought, herbivory, galls, fungal diseases, and demographic factors (Aldrich and Cavender-Bares 2011), although documentation specific to Shumard Oak was not found in the literature. The relative importance of these factors has not been determined (Aldrich et al. 2005b).
Oak Wilt is a caused by the fungus Ceratocystis fagacearum (Bretz) Hunt. Almost all species and varieties of oak are susceptible, but the red oaks are the most severely affected and usually die within the first three months of infection, although some Shumard Oaks may survive up to a year (CABI and EPPO 2011, USDA 2009). The disease develops in the sapwood and eventually fills the vascular tissues, restricting the flow of sap. The fungus commonly spreads from infected trees to healthy trees through contact between roots. This allows the disease to quickly spread to the entire stand (Canadian Forest Service 2012a), especially with the red oaks, which have shared root systems (Cook 2012). The disease also spreads by attaching fungal spores to boring and sap-feeding insects, including the Two-lined Chestnut Borer (Canadian Forest Service 2012a), through contaminated pruning tools, and through movement of infected plant material, including firewood (Cook 2012). The most visible symptoms are browning and wilting of the leaves, usually beginning in May. Eventually the entire crown is wilted (Canadian Forest Service 2012c) and the leaves are rapidly dropped, often within a three week period (Cook 2012). Distinctive black, elliptical spore mats form under the bark. Under the right environmental conditions, the mats produce a blister-like pressure pad that lifts and splits the bark, allowing access to insects (Cook 2012). Cook (2012) explains how to distinguish oak wilt from other diseases and summarizes prevention and management techniques.
Oak Wilt is widespread in the eastern US and has reached epidemic proportions in Minnesota, Wisconsin, Michigan, Ohio, Pennsylvania, and other mid-west states (USDA 2009). It has not yet been found in Ontario, but the province is considered at risk because of the proximity to infected US populations and the cross-border trade in plants and wood products that could carry the fungus or infected bark beetles (Canadian Forest Service 2012a).
Sudden Oak Death is a fatal disease of oaks caused by Phytophthora ramorum, an exotic oomycete (a fungus-like microorganism) that also causes non-lethal leaf blights, stem cankers, and tip dieback on a wide variety of trees, shrubs, vines, and herbaceous plants. In oaks, the disease manifests as red-brown to black trunk cankers that seep dark black to red or amber sap. Cankers may range in size from a few cm in diameter to three metres in length. Death occurs two or more years after initial infection when the infection girdles the tree. The crown often appears healthy throughout the infection and up to a year after the tree is effectively girdled, but turns completely brown within two to four weeks when the tree finally dies. Only oaks in the red oak group are susceptible to the disease (Davidson et al. 2003). Goheen et al. (2006) and Talgø and Chastagner (2012) describe the dispersal mechanisms for P. ramorum.
Shumard Oak does not occur naturally in any of the areas where P. ramorum has become established, so the susceptibility of this species to the disease is unknown. However, Widmer et al. (2012) demonstrated that other eastern North American oak species, including Pin Oak and Red Oak, are susceptible. P. ramorum has also naturally infected planted Red Oak trees in the Netherlands (Steeghs and de Gruyter 2006). Since Shumard Oak is known to hybridize with these species, it is likely also susceptible.
Sudden Oak Death was first observed in coastal California in the 1990s. By the end of 2004, the pathogen had killed tens of thousands of oaks in California, had become established in southern Oregon, and had been found on nursery stock and some plantings in 21 states and British Columbia (Tjosvold et al. 2005). The Canadian Food Inspection Agency (CFIA) collected P. ramorum from over 30 sites in British Columbia, including retail and wholesale nurseries, between 2003 and 2008 (Goss et al. 2011). However, Kristjansson and Miller (2009) rated the risk and consequences of infestation in Ontario as low.
Other pathogens, including other Phytophthora species, can cause similar symptoms so it is not possible to identify a P. ramorum infection only by disease symptoms. Laboratory diagnostic techniques must be used to determine whether Phytophthora is the cause of the disease and to identify the Phytophthora species (Neves and Maia 2012, Talgø and Chastagner 2012).
Population fragmentation and hybridization
Since Shumard Oak, like other oaks, is wind pollinated and produces large volumes of pollen, isolated trees are likely able to produce at least low quantities of seed (Knapp et al. 2000). However, Shumard Oak readily hybridizes with other red oaks (eFloras 2008). Isolated trees would therefore more likely be pollinated by other more abundant red oak species than by other Shumard Oaks, resulting in regeneration by hybrid oaks instead of genetically pure Shumard Oak.
Threats to urban trees
Shumard Oak has been planted as a shade or street tree in Windsor (Waldron et al. 1987). While the purpose of this management plan is not to protect ornamental trees, planted trees in urban settings could be a source of pollen or acorns for the enhancement and expansion of native populations. The genetic origin of planted urban trees needs to be determined. If they are cultivars they could potentially be "contaminating" the native gene pool. If they are from locally-sourced native stock and considered beneficial, threats to these urban trees may include (Clean Air Partnership 2012):
- increased competition for light, water and space to grow;
- physical limitations to growing space due to structures;
- inferior soils due to compaction, development and contamination;
- increased soil salinity due to road salt use;
- pest infestations;
- air pollution;
- physical damage due to human interference (e.g. pruning to protect buildings and infrastructure or for aesthetic reasons);
- poor drainage; and
- being subject to higher temperatures due to the urban heat island effect.
All of these factors cause stress, which makes trees more susceptible to infestation and mortality due insects and disease.
Climate change
Speer et al. (2009) studied a variety of oaks (but not Shumard Oak) in the southeastern United States, and suggest that moderate climate change impacts could expand the range and improve growth of oaks. A study by Inverson et al. (2008) supported this prediction, stating that oak species may benefit from the shifting climate and experience increases in suitable habitat. More specifically, climate change conditions such as wind and ice damage, hurricane intensity and frequency, extreme precipitation events, drought, forest fires, flooding, and warmer winter and warmer summer temperatures, may open forest canopies and aid oak regeneration (Inverson et al. 2008). However, frequent extreme weather events may not have these beneficial results in the longer term. Speer et al. (2009) suggest that the drought conditions created by climate change, will negatively impact oak growth. This would be especially true for Shumard Oak, which prefers wetter conditions.
Climate change will also influence the pest and disease pressures on tree species. Some pests are expected to flourish due to climate change, experiencing faster rates of development and movement beyond traditional ranges (Dukes et al. 2008, Inverson et al. 2008). The same is true of a variety of diseases, such as Oak Wilt, which benefits from warm and dry conditions (Boland et al. 2004). Stressful conditions such as drought will make trees more susceptible to disease and pest infestation (Boland et al. 2004). Though as Kliejunas (2011) notes, not all diseases will increase. Climate change impacts are specific to the type of disease and many diseases require wet conditions.
It should be noted that there are high levels of uncertainty associated with climate change predictions and modeling, and this is expressed throughout the literature.
4.0 Management
4.1 Goals and objectives
The management goal is to maintain or enhance self-sustaining populations of Shumard Oak within its current range in Ontario. Table 3. Management objectives
| No. | Management Objective |
|---|---|
| 1 | Protect and manage habitat to maintain extant populations in Ontario. |
| 2 | Support efforts to research and control the spread of diseases and pests considered to be high- priority threats to Shumard Oak. |
| 3 | Build capacity to accurately identify Shumard Oak. |
| 4 | Determine the current distribution and abundance of Shumard Oak populations in Ontario through inventory and monitoring. |
| 5 | Prepare and disseminate information on best management practices for Shumard Oak. |
| 6 | Support and implement landscape- and ecosystem-based planning and recovery initiatives to increase the amount of available habitat for Shumard Oak, and to enhance habitat connectivity for dispersal and population expansion. |
4.2 Management actions completed or underway
Measures to control European Gypsy Moth populations at small and large scales are well known and are available on many Ontario municipal and government websites. Short-term, localized methods include scrapping and destroying egg masses, trapping adults and larvae and spraying individual trees. Short-term, large scale methods of responding to severe outbreaks include aerial spraying of insecticides, release of sterile adults (to prevent reproduction) and biological controls such as fungal diseases specific to gypsy moths. There are long-term measures to prevent introduction, establishment and spread of the European Gypsy Moth into new areas. These include monitoring and inspection of travelers, monitoring and inspection of wood commodities (particularly nursery stock), silvicultural control methods, biological control, research into more effective control and eradication measures, and education. Media support is important if control measures are to be successful (Krcma-Nozic et al. 2000). Both gypsy moth species are subject to quarantine and are under surveillance by the Canadian Food Inspection Agency (CFIA). The CFIA has conducted annual gypsy moth surveys since 1954. Because infestations can cover large areas, several U.S. states and Canadian provinces including Ontario, have developed aerial control programs (Canadian Forest Service 2012b). Aerial spraying was conducted in Toronto and other southwestern Ontario cities in 2007 and 2008 to combat severe infestations (The Hamilton Spectator 2008, Hart 2012). Aerial spraying is proposed for Toronto in 2013 (Hart 2012).
Locally sourced Shumard Oak is grown by Ontario native plant nurseries and used in tree planting projects in Essex County (Gartshore pers. comm. 2012). The City of Windsor has planted Shumard Oak along urban streets to replace trees killed by Emerald Ash Borer (Waldron pers. comm. 2012). SCRCA has worked with Trees Ontario and Ontario tree nurseries to collect and grow local Shumard Oak seeds for restoration programs in Chatham-Kent, Lambton and Middlesex Counties (Mills and Craig 2008, SCRCA 2011).
ERCA collects Shumard Oak seeds at Maidstone Conservation Area, Devonwood Conservation Area, and some private properties in Essex County. The trees are grown in nurseries until they are ready for planting. Since 2005, ERCA has planted over 39,000 locally sourced Shumard Oak for restoration projects on private property and at ERCA owned properties. The oaks may not all be pure Shumard Oaks; ERCA's priority is a red oak that grows well on wet clay (Davies pers. comm. 2012).
SCRCA conducted a species at risk inventory of SCRCA managed lands in 2007 and 2008. Shumard Oak was found in two conservation areas in Lambton County. At both sites, regeneration appeared to be lacking. Most of the specimens were quite large, with flared butts and epicormic branching (Mills and Craig 2008). Shumard Oak was also found during 2006 surveys of the Bowens Creek Lands, which SCRCA manages on behalf of Lambton County. The Bowens Creek Lands Management Plan includes wetland creation/enhancement and reforestation (SCRCA 2010). This site and a nearby Ducks Unlimited Canada project are in the same block as Bickford Oak Woods Conservation Reserve, another known Shumard Oak site. In 2011, SCRCA created a five hectare wetland on the Bowens Creek Lands (SCRCA 2011). The Bowens Creek and Ducks Unlimited wetlands will likely help maintain or enhance the hydrologic regime preferred by Shumard Oak. SCRCA includes activities to promote Shumard Oak regeneration and address threats to the species in its forest management programs (Payne, pers. comm. 2012). The Bickford Oak Woods Conservation Reserve’s population was threatened by erosion and trampling caused by unauthorized ATV and dirt bike trails. The conservation reserve’s 2009 management plan prohibits the use of motorized vehicles.
In 2003, Parks Canada Agency (PCA) sponsored a SAR inventory of their property at "The Lakeshore" in Niagara, during which two mature Shumard Oak trees were reported and geo-referenced (Jalava 2004). The associated report makes management recommendations for the property, in which trees should be afforded a degree of protection. Additional surveys of "The Lakeshore" during the Environmental Assessment for Niagara-on-the-Lake wastewater servicing found eight young (non- reproducing) trees that may be hybrids of Shumard Oak and Pin Oak (Q. x. mutabilis).
The leaves had some characteristics of Shumard Oak and some characteristics of Pin Oak. The trees were re-visited in the fall, but since they were too young to produce acorns, identification could not be clarified (North-South Environmental 2011).
Shumard Oak is a nested conservation target of the Niagara River Corridor Conservation Action Plan (CAP) (Jalava et al. 2012) and the Essex Forests and Wetlands CAP (EFW NACP/CAP Team 2009).
Shumard Oak is included in the Wetland Plant List for the Ontario Wetland Evaluation System (OWES) as an indicator species for swamps in southern Ontario. This is the only plant list that can be used to delineate wetland boundaries using OWES. In addition, for each species at risk and provincially significant species observed during the evaluation, a completed NHIC Rare Species Reporting Form (or sufficient information to complete one) must be submitted with the wetland evaluation file (Ministry of Natural Resources 2013). Consequently, wetland evaluators should be aware of the species, actively searching for it while conducting wetland evaluations in southern Ontario, and documenting and reporting their observations.
Shumard Oak is known to occur in the provincially significant Kent-Elgin Shoreline Life Science Area of Natural and Scientific Interest (ANSI). Much of the woodland of this ANSI is within the Clear Creek Forest Provincial Nature Reserve (Ontario Parks 2003). The Park Management Plan for Clear Creek Forest Provincial Nature Reserve was completed in 2012. Although the management plan does not include specific recommendations relating to Shumard Oak, it does include provisions that address many of the threats to Shumard Oak and provide a mechanism to fill some of the knowledge gaps, including (OMNR 2012b):
- manage invasive species
- maintain natural shoreline
- restore vegetation communities
- improve and restore natural stream flow to Clear Creek
- prohibit use of motorized vehicles (ATVs)
- protect plant communities from trampling and other adverse impacts
- manage species at risk
- manage deer populations, and
- conduct inventory and monitoring programs for species at risk and vegetation restoration activities.
The CFIA maintains a publicly available list of plants and areas regulated for Phytophthora ramorum and other exotic plant pests and diseases (CFIA 2012).
4.3 Management plan approaches for action
Table 4. Management plan approaches for action for the Shumard Oak in Ontario
1. Objective: Protect and manage habitat to maintain extant populations in Ontario.
| Management Theme | Management Approach | Relative Priority | Threats or Knowledge Gaps Addressed | Relative Timeframe |
|---|---|---|---|---|
| Management | 1.1 Incorporate Shumard Oak into guidelines for managing forests in southern Ontario. | necessary |
|
short-term |
| Research, Protection | 1.2 Implement short term (e.g., five-year) intensive Significant Woodland and Provincially Significant Wetland identification program.
|
necessary |
|
short-term |
| Education | 1.3 Continue to train and certify new wetlands evaluators; increase the number of courses offered annually. | beneficial |
|
ongoing long-term |
| Management | 1.4 Install long term deer exclosures around Shumard Oaks in areas with hyper-abundant deer populations to encourage natural regeneration. | beneficial |
|
short-term |
| Research, Management | 1.5 Investigate impact of forest structure changes caused by Emerald Ash Borer on Shumard Oak regeneration. Support application of silvicultural techniques to promote Shumard Oak regeneration in impacted forests if necessary. | beneficial |
|
long-term |
| Protection, Communication | 1.6 Work with the responsible agencies to ensure that the design, construction, and maintenance of drains under the Drainage Act and the Tile Drainage Act and associated regulations protect Shumard Oak and other species at risk. | critical |
|
long-term ongoing |
2. Objective: Support efforts to understand and control the spread of pests and diseases considered to be high-priority threats to Shumard Oak.
| Management Theme | Management Approach | Relative Priority | Threats or Knowledge Gaps Addressed | Relative Timeframe |
|---|---|---|---|---|
| Outreach and Education | 2.1 Develop and implement effective media campaigns to educate public about threats of exotic pests and about prevention measures. Evaluate campaigns and use lessons learned to build more effective future campaigns. | critical |
|
ongoing, long-term |
| Monitoring | 2.2 Continue to support gypsy moth population monitoring and outbreak prediction programs. | necessary |
|
ongoing, long-term |
| Management | 2.3 Continue to support gypsy moth population control measures at all scales (local to international). | necessary |
|
ongoing, long-term |
| Research, Education and Outreach, Communications | 2.4 Build capacity to identify Agrilus (borer) species in Ontario. | beneficial |
|
long-term |
| Research, Management | 2.5 Determine impacts of Agrilus (borer) species and other insects on Shumard Oak; develop and implement control measures if necessary. | necessary |
|
long-term |
| Management | 2.6 Implement and support measures to prevent establishment and spread of exotic diseases (e.g. Sudden Oak Death). | necessary | Prevention and control of introduced diseases. | long-term |
| Monitoring and Assessment | 2.7 Continue to support ongoing broad-leaved tree pest monitoring programs in southern Ontario (e.g., EAB, Asian Longhorn Beetle, Gypsy Moth). Ensure that known Shumard Oak are included in these programs. Ensure field staff are able to identify Shumard Oak. | critical |
|
long-term |
| Research | 2.8 Support research into impacts of exotic earthworms on oaks and potential control measures. | necessary |
|
long-term |
| Research, Monitoring and Assessment | 2.9 In areas with high oak pest populations, monitor Shumard Oak to determine impacts. Develop monitoring protocol, including ecology based pest population thresholds for implementing monitoring program. Document impacts on survival, reproduction, and regeneration of Shumard Oak. Use larval galleries on Shumard Oak to determine which borer species attacked first and which species are secondary pests. |
beneficial |
|
long-term |
| Research | 2.10 Investigate pesticides and other measures to protect individual Shumard Oaks from insect pests and disease. | necessary |
|
ongoing long-term |
| Management, Stewardship | 2.11 Develop pest and disease intervention guidelines for Shumard Oak. Implement when necessary to maintain reproducing populations of Shumard Oak. Prioritize groups of trees in order to maintain potential for natural regeneration. | necessary |
|
ongoing short-term |
| Education and Outreach, Communications | 2.12 Educate recreational anglers and bait industry about the threat earthworms pose to Ontario forests and about proper methods to dispose of unused earthworm bait. Develop and distribute educational materials (e.g., add to Baitfish Primer, annual Fishing Regulations and other fishing related publications). Review effectiveness and revise as necessary to obtain significant reduction in # of anglers who report releasing live worms | beneficial |
|
ongoing long-term |
| Research, Inventory and Monitoring, Education and Outreach | 2.13 Implement the recommendations in Evers et al. (2012) to address the threats and impacts of exotic earthworms. | necessary |
|
long-term ongoing |
| Protection | 2.14 Work with federal government and other provincial agencies to prevent the establishment and dispersal of exotic species in ship ballast and through commodities. | critical |
|
long-term ongoing |
3. Objective: Build capacity to accurately identify Shumard Oak.
| Management Theme | Management Approach | Relative Priority | Threats or Knowledge Gaps Addressed | Relative Timeframe |
|---|---|---|---|---|
| Inventory, Monitoring and Assessment, Research | 3.1 Support research into identifying genetic markers for red oaks. | necessary |
|
long-term |
| Inventory, Monitoring and Assessment | 3.2 Build capacity to conduct genetic testing in Ontario; support training of genetics laboratory staff. Increase volume of plant specimens that can be sampled in Ontario by supporting expansion of government and private labs. | necessary |
|
long-term |
| Education and Outreach, Communications | 3.3 Develop oak identification certification program for foresters and related professionals (similar to butternut health assessor program). Train forestry professionals to identify oaks; administer competence tests and certify those who demonstrate high level of proficiency in accurately identifying oaks. Maintain list of certified oak identifiers. |
critical |
|
long-term |
4. Objective: Determine the current distribution and abundance of Shumard Oak populations in Ontario through inventory and monitoring.
| Management Theme | Management Approach | Relative Priority | Threats or Knowledge Gaps Addressed | Relative Timeframe |
|---|---|---|---|---|
| Inventory, Monitoring and Assessment | 4.1 Inventory and monitor known and historic sites. Conduct population counts, including evidence of seedling recruitment and vegetative growth. Note evidence of pests, disease and other threats. Submit data to provincial repository. |
necessary |
|
short-term ongoing |
| Inventory | 4.2 Develop, implement, and maintain a system to track locations of planted SAR plants, including Shumard Oak, and the original sources of the planted trees.
|
critical |
|
short-term ongoing |
5. Objective: Prepare and disseminate information on best management practices for the species.
| Management Theme | Management Approach | Relative Priority | Threats or Knowledge Gaps Addressed | Relative Timeframe |
|---|---|---|---|---|
| Education and Outreach; Communications | 5.1 Educate landowners, forestry professionals, woodlot associations, and MNR staff about insect pests of Shumard Oak and other oaks. Develop and distribute communication materials (e.g., Best Management Practices). Encourage and facilitate reporting of insect damage and mortality of oak trees. Work with federal government to ensure MNR forestry and species at risk staff are quickly informed when new insects or diseases that affect oaks are discovered in Ontario, or when such diseases and insects are discovered in new areas. |
necessary |
|
ongoing |
| Monitoring and Assessment, Education and Outreach, Communications | 5.2 Work with federal government to develop, implement, promote, and maintain a centralized reporting system that the general public can easily find and use to report tree damage and mortality from insects and diseases. | necessary |
|
long-term |
| Management, Outreach, Communications, Inventory and Monitoring | 5.3 Promote and monitor the use of locally-sourced native seed stock with nurseries, landscaping companies and agencies involved in tree planting initiatives (e.g., municipalities, conservation authorities, ENGOs). | necessary |
|
ongoing |
6. Objective: Support and implement landscape- and ecosystem-based planning and recovery initiatives to increase the amount of available habitat for Shumard Oak, and to enhance habitat connectivity for dispersal and population expansion.
| Management Theme | Management Approach | Relative Priority | Threats or Knowledge Gaps Addressed | Relative Timeframe |
|---|---|---|---|---|
| Management, Outreach, Communications | 6.1 Support initiatives that protect, restore and rehabilitate natural landscape connectivity in areas where Shumard Oak is found (e.g., municipal natural heritage systems plans, Carolinian Canada conservation action plans, conservation authority watershed plans). | necessary |
|
ongoing |
| Research, Stewardship, Education and Outreach | 6.2 Investigate short and long term impacts of EAB and other forest pests on woodlot owner attitudes and activities within the range of Shumard Oak. Support and implement programs to encourage woodlot retention and management within the range of Shumard Oak | critical |
|
long-term |
| Protection, Education and Outreach, Stewardship | 6.3 Continue to support the Conservation Land Tax Incentive Program and the Managed Forest Tax Incentive Program. Develop, and implement effective communications materials. Use a variety of communication platforms to make woodlot owners within the range of Shumard Oak aware of these programs. Assist interested landowners with the development and implementation of property plans. Monitor compliance with approved plans. Evaluate and monitor effectiveness of communications materials (e.g., through number of plans completed) and revise as needed. | critical |
|
long-term ongoing |
Glossary
Advanced reproduction or regeneration: regeneration that occurs via saplings and seedlings already present in the understory before canopy openings are created instead of by suckering or seeds that sprout after the canopy is opened (fw.ky.gov/forestrg.asp).
Apex: the free end of a leaf or leaf lobe; the tip of the leaf or lobe that is not the point of attachment.
Cap scar: the circular, light coloured mark on the flattened end of the acorn nut where the nut was attached to the cap. Also called the cup scar (Ward 1892).
Capillary water: water that is held to soil particles by soil tension. Plants use this water (Fraser and Fleming 2001).
Committee on the Status of Endangered Wildlife in Canada (COSEWIC): the committee responsible for assessing and classifying species at risk in Canada.
Committee on the Status of Species at Risk in Ontario (COSSARO): the committee established under section 3 of the Endangered Species Act, 2007 that is responsible for assessing and classifying species at risk in Ontario.
Conservation status rank: a rank assigned to a species or ecological community that primarily conveys the degree of rarity of the species or community at the global (G), national (N) or subnational (S) level. These ranks, termed G-rank, N-rank and S-rank, are not legal designations. The conservation status of a species or ecosystem is designated by a number from 1 to 5, preceded by the letter G, N or S reflecting the appropriate geographic scale of the assessment. The numbers mean the following:
1 = critically imperilled
2 = imperilled
3 = vulnerable
4 = apparently secure
5 = secure
NR = unranked - conservation status not yet assessed
U = unrankable - currently unrankable due to lack of information or due to substantially conflicting information about status or trends.
Coppicing: the practice of periodically cutting trees or shrubs back to ground level to stimulate growth (Oxford University Press 2008).
Denitrifying bacteria: bacteria that remove nitrates and nitrites from the soil. Desiccation: moisture loss or removal (Oxford University Press 2008).
Endangered Species Act, 2007 (ESA): the provincial legislation that provides protection to species at risk in Ontario.
Gravitational water: loose or excess water that replaces the air in soil pores. This type of groundwater is not accessible to plant roots and leads to plant drowning (Fraser and Fleming 2001).
Mycorrhyzal fungus: a fungus that forms a symbiotic relationship in and on the roots of host plants (McGraw-Hill 2002).
Oomycete: an organism that, like fungi, exhibits a filamentous growth habit, obtains nutrition by absorption, and reproduces via spores, but is more closely related to algae and green plants than to fungi. Also known as water molds, oomycetes cause seedling blights, damping-off, root rots, foliar blights and downy mildews, including some of the most devastating plant diseases: late blight on potato (cause of the Irish Potato Famine), downy mildew on grape vine, sudden oak death and soybean root and stem rot (Fry and Grünwald 2010).
Pericarp: the part of a fruit formed from the outer wall of the ovary (Oxford University Press 2008). In oaks, the hard outer skin of the acorn (Bonner and Vozzo).
Recalcitrant seeds: seeds that require relatively high moisture levels (25 to 45%) and therefore cannot be stored below freezing (Bonner 2008b).
Sapwood: the outer layer of wood in a tree. This is the living wood tissue that conducts liquid upwards (Waldron 2003).
Species at Risk Act (SARA): the federal legislation that provides protection to species at risk in Canada. This act establishes Schedule 1 as the legal list of wildlife species at risk to which the SARA provisions apply. Schedules 2 and 3 contain lists of species that at the time the Act came into force needed to be reassessed. After species on Schedule 2 and 3 are reassessed and found to be at risk, they undergo the SARA listing process to be included in Schedule 1.
Species at Risk in Ontario (SARO) List: the regulation made under section 7 of the Endangered Species Act, 2007 that provides the official status classification of species at risk in Ontario. This list was first published in 2004 as a policy and became a regulation in 2008.
Temperate-recalcitrant seeds: recalcitrant seeds that can be stored at or slightly below freezing. Genera with temperate-recalcitrant seeds include buckeye (Aesculus spp.), chestnut (Castanea spp.), oak (Quercus spp.), and redbay (Persea spp.). Some, but not all of these species can be stored for 3 to 5 years at near-maximum moisture contents (30 to 50%) and low temperatures (–3 to +4 °C) (Bonner 2008b).
References
Aldrich, P.R., and J. Cavender-Bares. 2011. Quercus. Pp. 89-129 in Kole C (ed) Wild Crop Relatives: Genomic and Breeding Resources. Springer, Berlin. xx+166 pp.
Aldrich, P.R., J.C. Glaubitz, G.R. Parker, O.E. Rhodes Jr. and C.H. Michler. 2005a. Genetic Structure Inside a Declining Red Oak Community in Old-Growth Forest. Journal of Heredity 2005:96(6):627–634.
Aldrich, P.R., G.R Parker, J. Romero-Severson, and C.H. Michler. 2005b. Confirmation of oak recruitment failure in Indiana old-growth forest: 75 years of data. Forest Science 51(5):406-416.
Anonymous. 2004. Navy Island Preliminary Survey. 4 pp.
Arny, N.P. 2006. Fact Sheet FOR 51: Common Oaks of Florida. School of Forest Resources and Conservation, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida, Gainesville, FL. 9 pp.
Atkinson, T.H., J.L. Foltz, R.C. Wilkinson, and R.F. Mizell. 2005. EENY-131: Asian Ambrosia Beetle, Granulate Ambrosia Beetle (no official common name), Xylosandrus crassiusculus (Motschulsky) (Insecta: Curculionidae: Scolytinae). Entomology and Nematology Department, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida, Gainesville, FL. 6 pp.
Boland, G.J., M.S. Melzer, A. Hopkin, V. Higgins and A. Nassuth. 2004. Climate change and plant diseases in Ontario. Canadian Journal of Plant Pathology, 26:335-350.
Bonner FT. 1982. The effect of damaged radicles of presprouted red oak acorns on seedling production. Tree Planters' Notes 33(4): 13–15.
Bonner, F.T. 2008a. Quercus L. oak. Pp. 928-938 in F.T. Bonner and R.P. Karrfalt (eds). 2008. Agriculture Manual 727 - Woody Plant Seed Manual. U.S. Department of Agriculture, Forest Service, Washington, DC.
Bonner, F.T. 2008b. Storage of Seeds. Pp. 85-96 in F.T. Bonner and R.P. Karrfalt (eds). 2008. Agriculture Manual 727 - Woody Plant Seed Manual. U.S. Department of Agriculture, Forest Service, Washington, DC.
Bonner, F.T. and J.A. Vozzo. 1987. Seed Biology and Technology of Quercus. US Forest Service General Technical Report SO-66. Southern Forest Experiment Station, Forest Service, US Department of Agriculture, New Orleans, LA. 34 pp.
Buckley, D.S. and R.M. Evans. 2004. Effect of season of cutting on the production and growth of White and Shumard Oak stump sprouts in Tennessee. Pp. 45-51 in D.A. Yaussy, D.M. Hix, R.P. Long, and P.C. Goebel (eds.). 2004. General Technical Report NE-316. Proceedings of the 14th Central Hardwoods Forest Conference. U.S. Department of Agriculture, Forest Service, Northeastern Research Station, Newtown Square, PA. 539 pp.
Bull, E.L. and J.A. Jackson. 2011. Pileated Woodpecker (Dryocopus pileatus). In Poole, A. (ed.). The Birds of North American Online. Cornell Lab of Ornithology, Ithaca, NY. Website: http://bna.birds.cornell.edu/bna/species/148 [Accessed 7 April 2013]
CABI and EPPO. 2011. Data Sheets on Quarantine Pests: Ceratocystis fagacearum and its vectors. 6 pp.
Cameron, E.K., E.M. Bayne, and M.J. Clapperton. 2007. Human-facilitated invasion of exotic earthworms into northern boreal forests. Ecoscience 14(4): 482-490
Canada – United States – Ontario - Michigan Border Transportation Partnership. 2008. Natural Heritage Impact Assessment, Recommended Plan, December 2008. 200 pp. Website: http://www.partnershipborderstudy.com/pdf/NaturalHeritageImpactAssessment_RecommendedPlan.pdf [Accessed 11 April 2013].
Canadian Food Inspection Agency. 2012. Phytophthora ramorum - Sudden Oak Death. Web site: http://www.inspection.gc.ca/plants/plant-protection/diseases/sudden-oak-death/eng/1327587864375/1327587972647 [Accessed January 22, 2013]
Canadian Forest Service, Natural Resources Canada. 2011a. Rosy (pink) gypsy moth. Web site: http://tidcf.nrcan.gc.ca/insects/factsheet/1000100. [Accessed January 22, 2013].
Canadian Forest Service, Natural Resources Canada. 2011b. Winter moth. Web site: http://tidcf.nrcan.gc.ca/insects/factsheet/1000088. [Accessed January 22, 2013].
Canadian Forest Service, Natural Resources Canada. 2011c. Shumard Oak. Web site: http://tidcf.nrcan.gc.ca/trees/factsheet/454. [Accessed January 22, 2013].
Canadian Forest Service, Natural Resources Canada. 2012a. 4.1 Area within which Moderate to Severe Defoliation Occurs Including Area of Beetle-killed Trees by Insects and Province/Territory, 1975-2011. Web site: http://nfdp.ccfm.org/data/compendium/html/comp_41e.html#bookMark_Fa. [Accessed January 22, 2013].
Canadian Forest Service, Natural Resources Canada. 2012b. Gypsy moth. Web site: https://tidcf.nrcan.gc.ca/insects/factsheet/9506 [Accessed January 22, 2013].
Canadian Forest Service, Natural Resources Canada. 2012c. Oak wilt. Web site: http://tidcf.nrcan.gc.ca/diseases/factsheet/1000099. [Accessed January 22, 2013].
Ciesla, W.M. 2003. Agrilus biguttatus. Exotic Forest Pest Information System For North America Version 1.1. Web site: http://spfnic.fs.fed.us/exfor/data/pestreports.cfm?pestidval=154&langdisplay=en glish [Accessed: January 1, 2013].
Clean Air Partnership. 2012. Clean Air Council Urban Forestry Scan: Report to the Clean Air Council. Clean Air Partnership, Toronto, Ontario. 45 pp.
Cleland, E. 2013. Email correspondence with R. Donley. January 2013. Forest Health Technical Specialist, Ministry of Natural Resources, Aylmer.
Committee on the Status of Endangered Wildlife in Canada (COSEWIC). 1999.
COSEWIC assessment and update status report on the shumard oak Quercus shumardii in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. vi + 11 pp. Web site: www.sararegistry.gc.ca/status/status_e.cfm [Accessed: 2 November 2012]
Connor, K. 2004. Storing acorns. Native Plants Journal 5(2):161-166.
Cook, B. 2012. Oak Wilt in Michigan’s Forest Resource. Extension Bulletin E-3169. Michigan State University Extension, East Lansing, Michigan. 8 pp.
Davidson, J.M., S. Werres, M. Garbelotto, E.M. Hansen, and D.M. Rizzo. 2003. Sudden Oak Death and Associated Diseases Caused by Phytophthora ramorum. Plant Management Network Plant Health Progress. 23 pp.
Davies, R. pers. comm. 2012. Email correspondence with R. Donley. November 2012. Forestry Technician, Essex Region Conservation Authority, Essex, ON.
Davis, A.S., and D.F. Jacobs. 2004. First-year survival of northern red oak seedlings planted on former surface coal mines in Indiana. Pp. 480–502, in Barnhisel, R.I. (ed.). 2004. Proceedings of A joint Conference of American Society of Mining and Reclamation 21st Annual National Conference and 25th West Virginia Surface Mine Drainage Task Force Symposium. American Society of Mining and Reclamation, Lexington, KY.
Demers, C., A. Long, C. Latt, and E. Willcox. 2011. SS-FOR-12: Establishing and Maintaining Wildlife Food Sources. School of Forest Resources and Conservation Department, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida, Gainsville, FL. 5 pp.
Dukes, J.S., J. Pontius, D. Orwig, J.R. Garnas, V.L. Rodgers, N. Brazee, B. Cooke, K.A. Theoharides, E.E. Stange, R. Harrington, J. Ehrenfeld, J. Gurevitch, M. Lerdau, K. Stinson, R. Wick and M. Ayres. 2008. Responses of insect pests, pathogens, and invasive plant species to climate change in the forests of northeastern North America: What can we predict? Canadian Journal of Forest Research, 39:231-248.
Eckel, P.M. 2002. The mystery oak: Quercus shumardii Buckley at Buckhorn Island. Clintonia 17(4):8
Edwards, M.B. 1990. Quercus shumardii Buckl. Shumard Oak. Pp. 734-737, in Burns, R.M. and B.H. Honkala. Silvics of North America. Volume 2. Hardwoods. USDA Forest Service Agric. Handbook 654, Washington, D.C. Website http://www.na.fs.fed.us/pubs/silvics_manual/volume_2/quercus/shumardii.htm [Accessed 8 April 2013]
eFloras (2008). eFlora of North America. Missouri Botanical Garden, St. Louis, MO & Harvard University Herbaria, Cambridge, MA. Web site: http://www.efloras.org [Accessed: November 22, 2012]
EFW NACP/CAP (Essex Forests and Wetlands Natural Area Conservation Plan / Conservation Action Plan) Team. 2009. Essex Forests and Wetlands Conservation Action Plan. Carolinian Canada Coalition, London. X + 63 pp.
ERCA (Essex Region Conservation Authority). 2013. Forestry and Tree-planting. Web site: http://www.erca.org/watershed/forestry_and_treeplanting.cfm [Accessed: January 11, 2013].
Evers, A.K., A.M. Gordon, P.A. Gray, and W.I. Dunlop. 2012. CCRR-23: Implications of a Potential Range Expansion of Invasive Earthworms in Ontario’s Forested Ecosystems: A Preliminary Vulnerability Analysis. Ontario Ministry of Natural Resources, Peterborough, Ontario. 31 pp.
Farrar, J.L. 1995. Trees in Canada. Fitzhenry and Whiteside Limited, Markham, Ontario, and the Canadian Forest Service, Natural Resources Canada, Ottawa, ON. x + 502 pp.
Filoso, G. and H. Larocque. 2010. Ecoregion profile: Lake Erie Lowland. Catalogue no. 16-002-X EnviroStats 4(1):8-12.
Frank, S., and S. Bambara. 2009. The Granulate Ambrosia Beetle. North Carolina State University, A&T State University Cooperative Extension. Available http://www.ces.ncsu.edu/depts/ent/notes/Ornamentals_and_Turf/trees/note111/note111.html. 3Pp.
Fraser, H. and R. Fleming. 2001. Environmental Benefits of Tile Drainage – Literature Review. University of Guelph, Guelph, Ontario. 25 pp.
Frelich, L.E., C.M. Hale, S. Scheu, A.R. Holdsworth, L. Heneghan, P.J. Bohlen, and P.B. Reich. 2006. Earthworm invasion into previously earthworm-free temperate and boreal forests. Biological Invasions 8:1235-1245.
Fry, William E. and Niklaus J. Grünwald. 2010. Introduction to Oomycetes. The Plant Health Instructor. DOI:10.1094/PHI-I-2010-1207-01
Garofalo, A. pers. comm. 2012. Email correspondence to R. Donley. December 2012. Consulting Ecologist.
Gartshore, M. pers. comm. 2013. Email correspondence to R. Donley. January 2013. St. Williams Forestry & Ecology Centre, St. Williams, ON.
Gartshore, M., pers. comm. 2012. Email correspondence to R. Donley. November 2012. St. Williams Forestry & Ecology Centre, S Williams, ON.
Gilman, E.F., and D.G. Watson. 1994. Fact Sheet ST-561 Quercus shumardii Shumard Oak. US Forest Service. 4Pp.
Goheen, E.M., E. Hansen, A. Kanaskie, N. Osterbauer, J. Parke, J. Pscheidt, and G. Chastagner. 2006. Sudden Oak Death and Phytophthora ramorum: A guide for forest managers, Christmas tree growers, and forest-tree nursery operators in Oregon and Washington. Oregon State University Extension Service. 18 pp.
Goheen, J.R., and R.K. Swihart. 2003. Food-hoarding behavior of gray squirrels and North American red squirrels in the central hardwoods region: Implications for forest regeneration. Canadian Journal of Zoology, 81:1636-1639.
Goss, E.M., M. Larsen, A. Vercauteren, Werres, S.; Heungens, K.; Grünwald, N.J. 2011. Phytophthora ramorum in Canada: Evidence for Migration Within North America and from Europe. Phytopathology 101(1):166-171. DOI: 10.1094/PHYTO-0510-0133
Guttman, S.I. and L.A Weigt. 1989. Electrophoretic evidence of relationships among Quercus (oaks) of eastern North America. Canadian Journal of Botany 67(2): 339-351.
Haack, R.A., and D.M. Benjamin. 1982. The biology and ecology of the twolined chestnut borer, Agrilus bilineatus (Coleoptera: Buprestidae) on oaks, Quercus spp., in Wisconsin. The Canadian Entomologist 114:385-396
Haack, R.A., D.M. Benjamin, and K.D. Haack. 1983. Buprestidae, Cerambycidae, and Scolytidae associated with successive stages of Agrilus bilineatus (Coleoptera: Buprestidae) infestation of oaks in Wisconsin. The Great Lakes Entomologist 16(2):47-55
Haack, R.A., T.R. Petrice, and J.E. Zablotny. 2009. First report of the European oak borer, Agrilus sulcicollis (Coleoptera: Buprestidae) in the United States. The Great Lakes Entomologist 42(1&2):1-7.
Hart, J. 2012. Staff report for action on Control of European Gypsy Moth Outbreak in the City of Toronto. October 22, 2012. Reference Number P:\2012\Cluster A\PFR\PE17-110912-AFS#16357. Parks, Forestry and Recreation, The Corporation of the City of Toronto. 8 pp.
Hendrix, P.F. and P.J. Bohlen. 2002. Exotic Earthworm Invasions in North America: Ecological and Policy Implications. BioScience, Vol. 52, No. 9, pp. 801-811
Hoebeke, E.R. 2010. Emerald Ash Borer and European Oak Borer: Two newly detected invasive wood-boring beetles threatening ash and oak trees in New York. Branching Out 17(3), 2 pp. insert.
Iverson, L., A. Prasad and S. Matthews. 2008. Modeling potential climate change impacts on the trees of the northeastern United States. Mitigation and Adaptation Strategies for Global Change, 13:487-516.
Jackson, J.A., and H.R. Ouellet. 2002. Downy Woodpecker (Picoides pubescens). In Poole, A. (ed.). The Birds of North American Online. Cornell Lab of Ornithology, Ithaca, NY. Web site: http://bna.birds.cornell.edu/bna/species/613 [Accessed 7 April 2013]
Jackson, L., T. Molet, and G. Smither. 2011. Exotic Wood Borer/Bark Beetle National Survey Guidelines. USDA-APHIS-PPQ-CPHST, Raleigh, NC. 264 pp.
Jalava, J.V. 2004. Species At Risk and Botanical Inventory of Parks Canada’s The Lakeshore and Paradise Grove Properties (Fort George National Historic Park, Niagara-on-the-Lake, Ontario). Parks Canada Agency, Ottawa. 38 pp.
Jalava, J.V., J. Baker, K. Beriault, A. Boyko, A. Brant, B. Buck, C. Burant, D. Campbell, W. Cridland, K. Frohlich, L. Goodridge, M. Ihrig, N. Kiers, D. Kirk, D. Lindblad, T. Van Oostrom, D. Pierrynowski, P. Robertson, M. L. Tanner, A. Thomson and T. Whelan. 2012. Niagara River Corridor Conservation Action Plan 2010 (updated Nov. 2012). Niagara River Corridor Conservation Action Planning Team and the Carolinian Canada Coalition. x + 63 pp.
Jendek, E., and V.V. Grebennikov. 2009. Agrilus sulcicollis (Coleoptera: Buprestidae), a new alien species in North America. The Canadian Entomologist 141:236-245.
Kartesz, J.T. 1999. A synonymized checklist and atlas with biological attributes for the vascular flora of the United States, Canada, and Greenland. First Edition. In: Kartesz, K.T. and C.A. Meacham (eds.).1999. Synthesis of the North American Flora, Version 1.0. North Carolina Botanical Garden. Chapel Hill, N.C.
Kirchoff BK, Leggett R, Her V, Moua C, Morrison J, Poole C. 2011. Principles of visual key construction - with a visual identification key to the Fagaceae of the southeastern United States. AoB Plants 2011: plr005; DOI:10.1093/aobpla/plr005. 48 pp.
Kliejunas, J.T. 2011. A Risk Assessment of Climate Change and the Impact of Forest Diseases on Forest Ecosystems in the Western United States and Canada. General Technical Report, The Forest Service of the United States Department of Agriculture, California, USA. 70 pp.
Knapp, E., K. Rice and M. Goedde. 2000. Habitat Fragmentation Limits Pollen Availability and Acorn Production in Blue Oak. Oaks, n Folks 15(1)
Krcmar-Nozic, E., B. Wilson, and L. Arthur. 2000. The potential impacts of exotic forest pests in North America: a synthesis of research. Information Report BC-X-387. Natural Resources Canada, Canadian Forest Service, pacific Forestry Centre, Victoria, BC.
Kristjansson, G.T. et Miller, S.J. 2009. Pest risk assessment of Phytophthora ramorum Werres, de Cock & Man in't Veld causal agent of Ramorum blight, Ramorum bleeding canker, Ramorum (shoot) dieback and Sudden Oak Death. Canadian Food Inspection Agency, PRA 2000-39 Version 4. 251 pp.
Krochmal, A. and C. Krochmal. 1982. Uncultivated nuts of the United States. Agriculture Information Bulletin 450. Washington, DC: U.S. Department of Agriculture, Forest Service. 89 p.
LaBonte, 2010. Eradication of an Exotic Ambrosia Beetle, Xylosandrus crassiusculus (Motschulsky), in Oregon. Pp. 41-43 in K. McManus and K.W. Gottschalk (eds). 2010. General Technical Report NRS-P-75: Proceedings 21st S. Department of Agriculture Interagency Research Forum on Invasive Species 2010. USDA Forest Service, Newtown Square, PA. 169 pp.
Linderman, R. G., de Sá, P. B., and Davis, E. A. 2007. Comparative susceptibility of plants native to the Appalachian range of the United States to inoculation with Phytophthora ramorum. Online. Plant Health Progress doi:10.1094/PHP-2007-0917-01-RS.
MacKenzie, A. 2013. Email correspondence with R. Donley. January 2013. Natural Heritage Education/Resource Management Supervisor, Pinery Provincial Park.
Marquis, D.A., P.L. Eckert, and B.A. Roach. 1976. Acorn Weevils, Rodents, and Deer All Contribute to Oak-Regeneration Difficulties in Pennsylvania. USDA Forest Service Research Paper NE-3561976 Northeastern Forest Experiment Station, Forest Service. U.S. Department Of Agriculture Upper Darby, PA. 5Pp.
Marquis, R.J. and J.T. Lill. 2010. Impact of plant architecture versus leaf quality on attack by leaf-tying caterpillars on five oak species. Oecologia 163(1): 203-213. doi:10.1007/s00442-009-1519-2
McGraw-Hill. 2002. McGraw-Hill Dictionary of Scientific and Technical Terms, 6th edition. The McGraw-Hill Companies, Inc. 2380 pp.
Meyers, G. 1984. Some notes on Shumard’s Oak in the Hamilton-Niagara region. The Wood Duck 37(9): 141-148.
Mills, C. and D. Craig. 2008. Woodland Species at Risk Inventory in the St Clair Region Conservation Authority. St. Clair Region Conservation Authority, Strathroy, Ontario.
Morsink, W.A.G., and P.D. Pratt. 1984. Shumard Oak, Quercus shumardii, in Essex County, Ontario. The Canadian Field-Naturalist 98(4) pp. 470-478.
Natural Heritage Information Centre (NHIC). 2012. Element Summary Report for Quercus shumardii. Ontario Ministry of Natural Resources, Peterborough, Ontario. Web site: https://www.biodiversityexplorer.mnr.gov.on.ca/nhicWEB/nhicIndex.jsp. [Accessed: November 19, 2012].
NatureServe. 2012. NatureServe Explorer: An online encyclopedia of life [web application]. Version 7.1. NatureServe, Arlington, Virginia. Web site: http://www.natureserve.org/explorer. [Accessed: November 18, 2012].
Nealis, V.G., and S. Erb. 1993. A Sourcebook for Management of the Gypsy Moth. Forestry Canada, Ontario Region, Great Lakes Forestry Centre, Sault Ste. Marie, ON. 57 pp.
Nelson, G. H., G.C. Walters, Jr., R.D. Haines, and C.L. Bellamy. 2008. A catalog and bibliography of the Buprestoidea of America North of Mexico. The Coleopterists Society, Special Publ. No. 4: 1-274.
Neves, D. and C. Maia. 20120. Phytophthora on Quercus suber L. (cork oak). JKI Data Sheets - Plant Diseases and Diagnosis (10). Julius Kühn-Institut, Federal Research Centre for Cultivated Plants, Quedlinburg, Germany. DOI 10.5073/jkidspdd.2012.010
North-South Environmental Inc. 2011. Niagara-On-The-Lake Wastewater Servicing, Harmonized Federal/Provincial Environmental Assessment - Terrestrial Natural Features. Campbellville, ON.
Ober, H.K. 2011. WEC248: The Value of Oaks to Wildlife. Wildlife Ecology and Conservation Department, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida, Gainesville, FL. 6 pp.
Oldham, M.J. 2010. Checklist of the Vascular Plants of Niagara Regional Municipality Ontario. Pp. 9-1 to 9-233. in Niagara Region Conservation Authority. 2010. Natural Areas Inventory 2006-2009 vol. 2. 223 pp.
Olson, D.F., Jr. 1974. Quercus L. oak. Pp. 692-703 in: C.S. Schopmeyer (ed.) Agriculture Handbook 450 - Seeds of woody plants in the United States. U.S. Department of Agriculture - Forest Service, Washington, DC
Ontario Biodiversity Council (OBC). 2010b. State of Ontario’s Biodiversity 2010. A report of the Ontario Biodiversity Council, Peterborough, Ontario [available: http://www.ontariobiodiversitycouncil.ca/index.php/reports].
Ontario Biodiversity Council. 2011. Ontario’s Biodiversity Strategy, 2011: Renewing Our Commitment to Protecting What Sustains Us. Ontario Biodiversity Council, Peterborough, ON.
Oxford University Press. 2008. The New Oxford American Dictionary, 2nd edition.
Oxford University Press, Inc. on Mac iOS 6.0.1.
OMNR (Ontario Ministry of Natural Resources). 1991. Rondeau Provincial Park Management Plan. Queen’s Printer for Ontario, Ontario, Canada. i + 31 pp
OMNR (Ontario Ministry of Natural Resources). 2012. Clear Creek Forest Park Management Plan. Queen’s Printer for Ontario. iii + 17 pp.
OMNR (Ontario Ministry of Natural Resources). 2013. Ontario Wetland Evaluation System, Southern Manual, 3rd edition, version 3.2. Queen’s Printer for Ontario. viii + 283 pp.
Ontario Parks. 2003. Clear Creek Forest: Terms of Reference. Queen’s Printer for Ontario. 10 pp.
Ontario Woodlot Association. 2003. Tree Conservation and Tree-cutting Bylaw Legislation in Ontario. OntarioWoodlot.com Woodland Notes. Ontario Woodlot Association, Kemptville, Ontario. 2 pp. Web site: http://ontariowoodlot.com/pdf_older/bylaw_leg_treecutting.pdf. [Accessed: 10 April 2013.
Oxford University Press. 2008. The New Oxford American Dictionary, 2nd edition.
Oxford University Press, Inc. on Mac iOS 6.0.1.
Patterson, W.A. 2006. The paleoecology of fire and oaks in eastern forests. Pp 2-19 in Dickinson, M.B. (ed.). 2006. Fire in Eastern Oak Forests: Delivering Science to Land Managers. Proceedings of a Conference November 15-17, 2005 Fawcett Center The Ohio State University, Columbus, Ohio. General Technical Report (GTR) NRS-P-1. United States Department of Agriculture, Forest Service, Northern Research Station, Newtown Square, PA.
Payne, T. pers. com. 2012. Personal communication with R. Donley. Dec 2012. Forest Management Technician, St. Clair Region Conservation Authority.
Peer, B.D. and E.K. Bollinger. 1997. Common Grackle (Quiscalus quiscula). In Poole, A. (ed.). The Birds of North American Online. Cornell Lab of Ornithology, Ithaca, NY. Web site: http://bna.birds.cornell.edu/bna/species/271 [Accessed 7 April 2013]
Porchuk, B., pers. comm. 2013. Correspondence with J.V. Jalava. January 2013. Ecological Consultant, Ontario.
Riccardi, C.L. B.C. McCarthy and R.P. Long. 2004. Oak seed production, weevil (Coleoptera: Curculionidae) populations, and predation rates in mixed-oak forests of southeast Ohio. Pp. 10-20 in Yaussy, D.A.; D.M. Hix, R.P. Long, and P.C. Goebel (eds.). 2004. Proceedings. 14th Central Hardwood Forest Conference. 2004 March 16-19; Wooster, OH. General Technical Report (GTR) NE-316. Newtown Square, PA: U.S. Department of Agriculture. Forest Service. Northeastern Research Station. 539 p
Schaefer, J.M., J. Cohen, and M.E. Hostetler. 2012. WEC 168: The Wood Duck. Wildlife Ecology and Conservation Department, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida, Gainesville, FL.
Schwert, D.P. 1977. The first North American record of Aporrectodea icterica (Savigny, 1826) (Oligochaeta, Lumbricidae), with observations on the colonization of exotic earthworm species in Canada. Canadian Journal of Zoology 55:245-248.
SCRCA 2010. Bowens Creek Lands Management Plan. St. Clair Region Conservation Authority, Strathroy, Ontario.
SCRCA. 2011. Authority working with Trees Ontario for seeds. Conservation Update October 2011:2.
Shackelford, C.E., R.E. Brown, and R.N. Conner. 2000. Red-bellied Woodpecker (Melanerpes carolinus). In Poole, A. (ed.). The Birds of North American Online. Cornell Lab of Ornithology, Ithaca, NY. Web site: http://bna.birds.cornell.edu/bna/species/500 [Accessed 7 April 2013]
Smith, K.G., J.H. Withgott, and P.G. Rodewald. 2000. Redheaded Woodpecker (Melanerpes erythrocephalus). In Poole, A. (ed.). The Birds of North American Online. Cornell Lab of Ornithology, Ithaca, NY. Web site: http://bna.birds.cornell.edu/bna/species/518 [Accessed 7 April 2013]
Soil Classification Working Group. 1998. The Canadian System of Soil Classification, 3rd ed. Agriculture and Agri-Food Canada Publication 1646. NRC Research Press, Ottawa, ON. 188 pp.
Speer, J.H., H.D. Grissino-Mayer, K.H. Orvis and C.H. Greenberg. 2009. Climate response of five oak species in the eastern deciduous forest of the southern Appalachian Mountains, USA. Canadian Journal of Forest Research, 39:507-518.
Steeghs, M.H.C.G, and J. de Gruyter. 2006. Phytophthora ramorum Experience and Approach in the Netherlands. Pp. 367-368 in: S.J. Frankel, P.J. Shea, and M.I. Haverty (eds). 2006. General Technical Report PSW-GTR-196. Proceedings of the Sudden Oak Death Second Science Symposium: the State of Our Knowledge. U.S. Department of Agriculture, Forest Service, Pacific Southwest Research Station, Albany, CA. 571 pp.
Sullivan, A.R., J.F. Lind, T.S. McCleary, J. Romero-Severson, and O. Gailing. 2012. Development and Characterization of Genomic and Gene-Based Microsatellite Markers in North American Red Oak Species. Plant Molecular Biology Reporter (online). http://dx.doi.org/10.1007/s11105-012-0495-6
Sullivan, J. 1993. Quercus shumardii. In: Fire Effects Information System, [Online]. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory (Producer). Web site: http://www.fs.fed.us/database/feis. [Accessed December 20, 2012].
Sutherland, W.J., S. Bardsley, L. Bennun, M. Clout, I.M. Côté, M.H. Depledge, L.V. Dicks, A.P. Dobson, L. Fellman, E. Fleishman, D.W. Gibbons, A.J. Impey, J.H. Lawton, F. Lickorish, D.B. Lindenmayer, T.E. Lovejoy, R.M. Nally, J. Madgwick, L.S. Peck, J. Pretty, S.V. Prior, K.H Redford, J.P.W Scharlemann, M. Spalding, and A.R. Watkinson, 2011. Horizon scan of global conservation issues for 2011. Trends in Ecology & Evolution 26:10-16.
Talgø, V. and G. Chastagner. 2012. Phytophthora on Abies spp. (true firs). JKI Data Sheets - Plant Diseases and Diagnosis (3). Julius Kühn-Institut, Federal Research Centre for Cultivated Plants, Quedlinburg, Germany. 14 pp. DOI: 10.5073/jkidspdd.2012.003
Tanentzap, A.J., D.R. Bazely, S. Koh, M. Timciska, E.G. Haggith, T.J. Carleton and D.A. Coomes. 2010. Seeing the forest for the deer: Do reductions in deer-disturbance lead to forest recovery? Biological Conservation, 144:376-382.
Tarvin, K.A., and G.E. Wooifenden. 1999. Blue Jay (Cyanocitta cristata). In Poole, A. (ed.). The Birds of North American Online. Cornell Lab of Ornithology, Ithaca, NY. Web site: http://bna.birds.cornell.edu/bna/species/469 [Accessed 7 April 2013]
The Hamilton Spectator. 2008. Gypsy moth spray program expands. Hamilton, ON. Apr 15 2008.
Tiunov, A.V., C.M. Hale, A.R. Holdsworth, T.S. Vsevolodova-Perel. 2006. Invasion patterns of Lumbricidae into the previously earthworm-free areas of northeastern Europe and the western Great Lakes region of North America. Biological Invasions 86:1223–1234
Tjosvold, S.A., K.R. Buermeyer, C. Blomquist, and S. Frankel. 2005. Division of Agriculture and Natural Resources Publication 8156: Nursery Guide for Diseases Caused by Phytophthora ramorum on Ornamentals: Diagnosis and Management. Regents of the University of California, Division of Agriculture and Natural Resources. 18 pp.
Trehane, P. 2010. The Oak Names Checklist. International Cultivar Registrar for the International Oak Society, the International Cultivar Registration Authority (ICRA) for the genus Quercus. Web site: http://www.oaknames.org. [Accessed December 29, 2012]
USDA. 2009. Plant Guide: Shumard Oak. National Plant Data Center and the Biota of North America Program, Natural Resources Conservation Service, United States Department of Agriculture, Davis, California. 4 pp.
USDA–APHIS. 2011. New Pest Response Guidelines: Exotic Wood-Boring and Bark Beetles. USDA–APHIS–PPQ–EDP-Emergency Management, Riverdale, Maryland. 256 pp.
Van Lear, D.H. 2004. Upland oak ecology and management. Pp. 65-71 in Spetich, Martin A., ed. 2004. Upland Oak Ecology Symposium: History, Current Conditions, and Sustainability. General Technical Report SRS–73. Asheville, NC: U.S. Department of Agriculture, Forest Service, Southern Research Station. 311 pp.
Van Wagner, R. 2013. Email correspondence with R. Donley. January 2013. Environmental Project Coordinator, Lower Thames Valley Conservation Authority.
Waldron, G. 1983. Shumard Oak (Quercus shumardii) new to Ontario. The Plant Press 1(3):54
Waldron, G.W. 1984. Status Report on the Shumard Oak Quercus shumardii in Canada. Committee on the Status of Endangered Wildlife in Canada. Canadian Wildlife Service, Ottawa, ON. Unpublished report. 22 pp.
Waldron, G.W., pers. comm. 2013a. Email correspondence to R. Donley. April 2013. Consultant, Gerry Waldron Consulting Ecologists, Amherstburg, ON.
Waldron, G.W., pers. comm. 2013b. Telephone conversation with R. Donley, January 2013. Consultant, Gerry Waldron Consulting Ecologists, Amherstburg,
Waldron, G.W., pers. comm. 2012. Email correspondence to R. Donley. November 2012. Consultant, Gerry Waldron Consulting Ecologists, Amherstburg,
Waldron, G.W. 2003. Trees of the Carolinian Forest: A Guide to Species, Their Ecology and Uses. Boston Mills Press, Erin, Ontario. 275 pp.
Waldron, G.W., S.W. Aboud, J.D. Ambrose and G.A. Meyers. 1987. Shumard Oak, Quercus shumardii, in Canada. The Canadian Field-Naturalist 101():532-538.
Waller, D.M. 2008. White-tailed deer impacts in North America and the challenge of managing a hyperabundant herbivore. Pp 135-147 in Gaston, A.J., T.E. Golumbia, J.L. Martin, and S.T. Sharpe (eds.) 2008. Lessons from the Islands: introduced species and what they tell us about how ecosystems work. Proceedings from the Research Group on Introduced Species 2002 Symposium, Queen Charlotte City, Queen Charlotte Islands, British Columbia. Canadian Wildlife Service, Environment Canada, Ottawa
Ward, H.M. 1892. The Oak: A Popular Introduction to Forest-botany. D. Appleton and Company, New York, NY. 175 pp.
Widmer, T.L., N. Shishkoff, and S.C. Dodge. 2012. Root susceptibility and inoculum production from roots of eastern United States oak species to Phytophthora ramorum. Plant Disease 96(11):1675-1682.
Wilson, B.F. 1968. Red maple stump sprout development the first year. Harvard Forest Paper 18.
Wisconsin Council on Forestry. 2013. Deer Impacts on Forest Ecology and Management. Website: http://council.wisconsinforestry.org/ [Accessed 7 April 2013]
Wisconsin Council on Forestry. 2006a. Deer - Impacts on Forest Ecology and Management: 2006 Natural Oak Regeneration Survey. Website: http://council.wisconsinforestry.org/deer/deerregen.php [Accessed 7 April 2013]
Wisconsin Council on Forestry. 2006b. Deer - Impacts on Forest Ecology and Management: 2006 Plantation (artificial regeneration) Assessment. Website: http://council.wisconsinforestry.org/deer/deerassess.php [Accessed 7 April 2013]
Woodliffe, P.A. 2013a. Email correspondence with R. Donley. January 2013. (Retired) District Biologist, Ministry of Natural Resources.
Woodliffe, P.A. 2013b. Comments on jurisdictional review draft. March 2013. (Retired) District Biologist, Ministry of Natural Resources.
Zablotny, J.E. 2009. Discriminating Agrilus sulcicollis Lacordaire from Agrilus cyanescens Ratzeburg. Animal and Plant Health Inspection Service, US Department of Agriculture. 2Pp. Website: http://caps.ceris.purdue.edu/screening/agrilus_sulcicollis_versus_a_cyanescen s [Accessed 24 January 2013]
Zammit, T. pers. comm. 2013. Telephone conversation with R Donley. January 2013. Aquatic and Terrestrial Ecologist, Grand River Conservation Authority, Cambridge, ON.
Zoetewey, C. pers. comm. 2013. Telephone conversation with R Donley. January 2013. Technical Services Operator, Niagara National Historic Sites, Parks Canada, Niagara-on-the-Lake, ON.
Appendix 1. Key identification features of Red Oaks found in Ontario
Habitat
| Feature | Shumard Oak (Quercus shumardii) | Black Oak (Quercus velutina) | Red Oak (Quercus rubra) | Hill’s Oak (Quercus eliipsoidalis) | Pin Oak (Quercus palustris) |
|---|---|---|---|---|---|
| General | Grows on moist, fine textured soils, usually near water (Farrar: on lowlands, along streams, on poorly drained, heavy soils) | Grows on well drained, coarse textured soils; sandy soils; also on steep slopes with heavy soils | Grows on mesic to xeric sites | Grows on well drained, coarse textured soils (eFloras: Dry sandy sites, rarely on moderately mesic slopes or uplands) | Grows on poorly drained soils, in swamps, and along streams; also grows on well-drained loamy soils |
| Ontario Waldron (2003) | Grows on clay lowland swamps and floodplains and on sand lowland swamps | Grows on moraines, sand and loam uplands, inland dunes and eskers, shore dunes, and acidic clay uplands | Grows on clay uplands, moraines, sand and loam uplands, inland dunes and eskers, and shore dunes | Grows on moraines, sand and loam uplands, and inland dunes and eskers | Grows on clay lowland swamps and floodplains and on sand lowland swamps, often poorer drainage than Shumard Oak sites |
Bark
| Feature | Shumard Oak (Quercus shumardii) | Black Oak (Quercus velutina) | Red Oak (Quercus rubra) | Hill’s Oak (Quercus eliipsoidalis) | Pin Oak (Quercus palustris) |
|---|---|---|---|---|---|
| Young | Young bark smooth, brownish green or light grey, becoming grey or greyish brown during first year | Young bark smooth, dark grey | Young bark smooth, dark grey | (information not available) | Young bark thin, smooth, greyish-brown |
| Mature colour | Mature bark dark grey | Mature bark very dark, almost black | Mature bark pale grey | Mature bark dark grey- brown | Mature bark greyish- brown, thin, smooth |
| furrows | Bark furrows shallow, dark brown (Farrar: bark deeply furrowed) | Bark furrows deep | Bark furrows shallow, dark, and wide | Bark furrows shallow | Bark furrows shallow, inconspicuous |
| ridges | Broken bark ridges, somewhat scaly | Bark ridges irregular, rounded, and broken into squarish segments | Continuous wide, flat, pale grey bark ridges | Narrow vertical plates | Mature bark divided into narrow inconspicuous ridges |
| Inner bark | Inner bark pinkish | Inner bark yellowish | Inner bark pinkish | Inner bark light yellow or orangeish | Inner bark reddish |
Form
| Feature | Shumard Oak (Quercus shumardii) | Black Oak (Quercus velutina) | Red Oak (Quercus rubra) | Hill’s Oak (Quercus eliipsoidalis) | Pin Oak (Quercus palustris) |
|---|---|---|---|---|---|
| Branches | Massive, ascending branches | Lower branches horizontal, upper branches ascending. Secondary branches droop to 90 degrees to main branch. | Massive branches. | Branches not massive, lower branches often descending. | Branches not massive, lower branches descending, centre branches horizontal, upper branches ascending. Numerous persistent dead lower branches and "pin-like" twigs. |
| Twigs | Twigs smooth, reddish or grey to greyish brown, slender. | Twigs dark reddish-brown, moderately stout, hairless or slightly hairy. | Twigs shiny reddish-brown, moderately stout. | Twigs bright or dark reddish-brown, slender, hairy when young, become smooth and hairless. | Twigs slender, hairless, reddish-brown. |
| Trunk | Trunk frequently buttressed (have roots that extend about the ground as a plate-like growth to support the tree), tapering to a trunk <1 m in diameter. | Trunk distinct up to the crown. | Trunk straight, not distinct into crown. | Lower trunk often has stubs of dead branches. | Trunk straight with a gradual taper, distinct well into the crown. |
Buds
| Feature | Shumard Oak (Quercus shumardii) | Black Oak (Quercus velutina) | Red Oak (Quercus rubra) | Hill’s Oak (Quercus eliipsoidalis) | Pin Oak (Quercus palustris) |
|---|---|---|---|---|---|
| Terminal buds | Terminal bids on vigorous twigs average < 6 mm long. | Terminal bids on vigorous twigs average < 6 mm long. | Terminal bids on vigorous twigs average < 6 mm long. | Terminal bids on vigorous twigs average < 6 mm long. | Terminal bids on vigorous twigs average < 6 mm long. |
| Bud hairs | Buds usually hairless or very slight downy, hairs grey or straw-coloured, can be nearly as hairy as Black Oak | Buds entirely covered with dense, greyish-white hairs. | Buds hairless except for a few brownish hairs at the tip. | Buds almost hairless, usually have silvery or tawny downy hairs at tip. | Buds almost hairless, may have hairs at tip. |
| Bud colour | Buds very pale grey-brown (Morsink and Pratt: terminal buds are dark brown with faintly hairy scales). | Buds appear grey due to dense hairs that completely obscure scale. | Buds shiny reddish-brown. | Buds brown. | Buds light chesnut-brown |
Leaves
| Feature | Shumard Oak (Quercus shumardii) | Black Oak (Quercus velutina) | Red Oak (Quercus rubra) | Hill’s Oak (Quercus eliipsoidalis) | Pin Oak (Quercus palustris) |
|---|---|---|---|---|---|
| Size | Leaves 8 to 18 cm long (up to 20 cm in southern US) | Leaves 10 to 20 cm long | Leaves 10 to 20 cm long | Leaves 7 to 16 cm long (usually 12 cm or less) | Leaves 7 to 15 cm long (relatively small) |
| Number of lobes | Leaves usually have 7 to 9 lobes, may have as few as 5 or as many as 11. | 5 to 7 lobes. | 9 to 11 loves (Farrar: 7 to 9 lobes) | 5 to 7 lobes. | 5 to 7 lobes (usually 5) |
| Colour | Leaves tend to be lustrous dark green above, paler beneath. | Leaves shiny dark green above, yellowish-brown and rough beneath. | Leaves tend to be dull yellowish green above, paler below. | Lower surface glossy light green, secondary veins raised on both surfaces. | Pale lustrous upper surface, lower surface paler (farrar: upper surface shiny dark green) |
| Retention | Leaves remain green after other oaks have turned brown; may be retained into winter | Leaves not retained into late fall or winter | Leaves not retained into late fall or winter | Dried leaves may be retained into winter | Leaves may be retained into winter |
| Hairs | Conspicuous hair tufts in vein axils on underside of leaf | Short hairs along veins and in vein axils on underside of leaf; young leaves velvety | Vein axils hairless or with less developed hair tufts | Faint hair tufts in vein axils on underside of leaf, otherwise hairless below | Faint hair tufts in vein axils on underside of leaf, otherwise hairless below |
| Sinuses | Deep, relatively narrow, rounded sinuses; indented ¾ of width of leaf, may extend to within a few mm of the midrib | Deep U-shaped sinuses; lobes have parallel sides | Shallow, wide, V-shaped sinuses (makes lobes taper from base to tip); sinuses rarely indented to ⅔ of leaf width | Deep, wide, sinuses, usually indented more than ½ the distance to the midrib; lobes often constricted towards the base | Deep, wide, U-shaped sinuses; basal (lower) lobes often recurved. |
| Form | Largest laterial loves of leaves growing in direct sunlight much longer than the leaf is wide between opposite sinuses | Central lobe may be twice as long as the width of the leaf between opposite sinuses. | Largest lateral lobes of leaves growing in direct sunlight are each as long as, or slightly longer than, the leaf is wide between opposite sinuses. | Largest lateral lobes of leaves growing in direct sunlight much longer than the leaf is wide between opposite sinuses | Largest lateral lobes of leaves growing in direct sunlight up to 3 times longer than the leaf wide between opposite sinuses. |
| Stalks | Leaf stalks 5 to 7 cm long (Waldron 2003: 2 to 6 cm long) | Leaf stalk stout, 7 to 15 cm long | Leaf stalk stout, 1 to 3 mm wide, 2.5 to 5 cm long |
Leaf stalk slender, 3.5 to 5 cm long |
Leaf stalk slender, 2 to 5 cm long |
Acorns
| Feature | Shumard Oak (Quercus shumardii) | Black Oak (Quercus velutina) | Red Oak (Quercus rubra) | Hill’s Oak (Quercus eliipsoidalis) | Pin Oak (Quercus palustris) |
|---|---|---|---|---|---|
| Size and shape | Large acorns, 15 to 30 mm long, usually 25 mm long | Small scorns, usually about 15 mm long; short-stalked or stalkless (Farrar: 12 to 20 mm long) | Large acorns, 17 to 30 mm long, usually 25 mm; bullet shaped (Farrar: 12 to 25 mm long) | Small acorns usually 15 mm long (Farrar: 12 to 18 mm long) | Small acorns, usually less than 18 mm long, hemispherical (almost as wide as long) Farrar: 9 to 13 mm long) |
| Cap scales | Acorn cap scales covered in a grey, woolly coating of dense bent matted hairs (tomentose), grey edged | Entire inner surface of acorn cap hairy; scales dull brown, loose-fitting, thin, and slightly hairy, may form a fringe to the cap | Acorn cap scales shiny, hairless, red edged | Scales reddish-brown, covered in very short, soft hairs; scales have straight or slightly concave margins | Acorn cap scaled tight-fitting, thin, pointed, hairy, and reddish-brown |
| Cap size and shape | Acorn cap shallow, saucer-shaped, covers about ⅓ of the nut | Acorn cap bowl shaped, covers about ½ of nut | Acorns either narrow-long with a deep, bowl-shaped cup covering ½ the nut, or as wide as long with a flat cup covering ¼ of the nut | Acorn cap tapers to a stalk-like base, is 10 to 15 mm wide, and covers ⅓ to ½ the nut | Acorns cap saucer-shaped, covering ¼ of the nut |
Flowers
| Feature | Shumard Oak (Quercus shumardii) | Black Oak (Quercus velutina) | Red Oak (Quercus rubra) | Hill’s Oak (Quercus eliipsoidalis) | Pin Oak (Quercus palustris) |
|---|---|---|---|---|---|
| Flowering time | Flowers appear in May with the leaves | Flowers appear in May when leaves are half grown | Flowers appear in May when leaves are half grown | (information not available) | Flowers appear in April when leaves only one-third grown |
| Catkins |
Slender, hanging, hairless catkins, 14 to 18 cm long |
Hairy, hanging catkins, 10 to 15 cm long |
Hairy catkins (length not available) |
Catkins 4 to 5 cm long | Hairy catkins, 5 to 7.5 cm long |
|
Female flowers |
Female flowers tiny; solitary or paired on short stems in the leaf axils | Female flowers inconspicuous; solitary or in pairs on short, hairy stems |
Female flowers inconspicuous; solitary or paired on short, hairless stems |
Female flowers inconspicuous |
Female flowers tiny and hairy; solitary or in pairs on short hairy stems |