Shagreen and Toothed Globe recovery strategy
Read the recovery strategy for the Shagreen and Toothed Globe, two snails at risk in Ontario.


About the Ontario recovery strategy series
This series presents the collection of recovery strategies prepared or adopted as advice to the Province of Ontario on the recommended approach to recover species at risk. The Province ensures the preparation of recovery strategies to meet its commitments to recover species at risk under the Endangered Species Act, 2007 (ESA) and the Accord for the Protection of Species at risk in Canada.
What is recovery?
Recovery of species at risk is the process by which the decline of an endangered, threatened, or extirpated (lives somewhere in the world, and at one time lived in the wild in Ontario, but no longer lives in the wild in Ontario) species is arrested or reversed, and threats are removed or reduced to improve the likelihood of a species’ persistence in the wild.
What is a recovery strategy?
Under the ESA a recovery strategy provides the best available scientific knowledge on what is required to achieve recovery of a species. A recovery strategy outlines the habitat needs and the threats to the survival and recovery of the species. It also makes recommendations on the objectives for protection and recovery, the approaches to achieve those objectives, and the area that should be considered in the development of a habitat regulation. Sections 11 to 15 of the ESA outline the required content and timelines for developing recovery strategies published in this series.
Recovery strategies are required to be prepared for endangered and threatened species within one or two years respectively of the species being added to the Species at risk in Ontario list. Recovery strategies are required to be prepared for extirpated species only if reintroduction is considered feasible.
What’s next?
Nine months after the completion of a recovery strategy a government response statement will be published summarizing the actions the Government of Ontario intends to take in response to the strategy. The implementation of recovery strategies depends on the continued cooperation and actions of government agencies, individuals, communities, land users, and conservationists.
For more information
To learn more about species at risk recovery in Ontario, visit the Ministry of the Environment, Conservation and Parks Species at risk webpage.
Recommended citation
Pivar, R.J. 2023. Nicolai, A. and Linton, J. Recovery Strategy for the Shagreen (Inflectarius inflectus) and Toothed Globe (Mesodon zaletus) in Ontario. Ontario Recovery Strategy Series. Prepared for the Ministry of the Environment, Conservation and Parks, Peterborough, Ontario. v + 37 pp.
Cover illustrations: Left photo: Inflectarius inflectus, photo by A. Nicolai (2013); right photo: Mesodon zaletus, UMMZ105034, recorded by C. Goodrich and M. L. Winslow in 1890 in Leamington, University of Michigan Museum of Zoology, Bryant Walker collection, photo by UMMZ Mollusk Division.
© King’s Printer for Ontario, 2023
ISBN 978-1-4868-6476-8 HTML
ISBN 978-1-4868-6477-5 PDF
Content (excluding illustrations) may be used without permission, with appropriate credit to the source.
Cette publication hautement spécialisée « Recovery strategies prepared under the Endangered Species Act, 2007 », n’est disponible qu’en anglais en vertu du Règlement 411/97 qui en exempte l’application de la Loi sur les services en français. Pour obtenir de l’aide en français, veuillez communiquer avec recovery.planning@ontario.ca.
Authors
Robert J. Pivar – Natural Resource Solutions Inc.
Annegret Nicolai – Living Lab CLEF/Université Rennes 1
Jessica Linton – Natural Resource Solutions Inc.
Acknowledgments
The authors would like to thank Tammy Dobbie and Melanie Lefaive of Point Pelee National Park for sharing their knowledge on species biology, current and potential future threats and for sharing data from their snail monitoring plots.
Declaration
The recovery strategy for Shagreen (Inflectarius inflectus) and Toothed Globe (Mesodon zaletus) was developed in accordance with the requirements of the Endangered Species Act, 2007 (ESA). This recovery strategy has been prepared as advice to the Government of Ontario, other responsible jurisdictions and the many different constituencies that may be involved in recovering the species.
The recovery strategy does not necessarily represent the views of all individuals who provided advice or contributed to its preparation, or the official positions of the organizations with which the individuals are associated.
The recommended goals, objectives and recovery approaches identified in the strategy are based on the best available knowledge and are subject to revision as new information becomes available. Implementation of this strategy is subject to appropriations, priorities and budgetary constraints of the participating jurisdictions and organizations.
Success in the recovery of this species depends on the commitment and cooperation of many different constituencies that will be involved in implementing the directions set out in this strategy.
Responsible jurisdictions
Ministry of the Environment, Conservation and Parks
Environment and Climate Change Canada – Canadian Wildlife Service, Ontario
Parks Canada Agency
Executive summary
Shagreen is a medium-sized land snail (adult shell width about 1 cm) with a shell that is depressed, yellow to brown in colour, with three tooth-like denticles in the shell opening and no hole in the middle of the shell where whorls come together. Toothed Globe is a large land snail (adult shell width 2.4 – 3.1 cm) with a globose-depressed, yellow, solid shell that has a tooth-like denticle in the opening. Both species are part of the unique fauna of the Carolinian Forest in Canada and may have significance for ecosystem function through nutrient cycling. The range edge population in Canada is important for the global conservation of both species.
In Ontario, Shagreen is extant in Essex County on Middle Island, and Pelee Island, both in Lake Erie. It appears to have been extirpated from southwestern Ontario mainland sites and two other Lake Erie islands. Toothed Globe appears to have been extirpated from most southwestern Ontario sites, including Lake Erie islands, but may still occur at historical sites in Essex and Middlesex counties. At present, there are no known extant locations.
Both Shagreen and Toothed Globe are currently listed as endangered under the Species at Risk in Ontario (SARO) list. Key threats for both species include climate change (droughts, changes in frost regimes), prescribed burns, and habitat modifications due to invasive species such as earthworms and hyperabundant Double-crested Cormorants, both of which affect leaf litter and soil conditions. Additionally, pollution and any direct and indirect human impacts (for example, habitat alteration, prescribed fire) specific to remaining sites, or potentially remaining sites in the case of Toothed Globe, threaten both species.
The recommended recovery goal for Shagreen is to maintain, and, where possible, support the natural expansion of the current subpopulations. The recommended recovery goal for Toothed Globe is to increase knowledge of the species and its habitat, and, if populations are found to exist, maintain and support the natural expansion of the subpopulations.
The recovery goals for these species are focused on addressing knowledge gaps, mitigating threats and enhancing habitat to allow for long-term population persistence and expansion in Ontario. To achieve these goals, recommended short-term recovery objectives are identified below:
- Engage government land managers, private landowners, and Indigenous communities in surveying suitable habitats to determine the current distribution of Shagreen in Ontario and whether Toothed Globe is still extant in the province.
- Assess and mitigate threats to all extant subpopulations, and, where appropriate, historical/presumed extirpated occurrence sites.
- Conduct and/or support research that fills knowledge gaps related to biology, population size, and habitat requirements that inform recovery efforts.
- Enhance and/or create habitat, where feasible and necessary, to increase habitat availability for extant subpopulations.
Information on the spatial limits of habitat used by Shagreen and Toothed Globe is lacking. When information on habitat and dispersal ability becomes available, the area prescribed as habitat could be described more precisely and should be revisited. It is recommended that all Ecological Land Classification (ELC) ecosites occupied by an extant subpopulation be prescribed as habitat in a habitat regulation. Specifically, the regulated area should be defined using a contiguous ecological area encompassing all occupied ecosites and any suitable unoccupied ecosites immediately adjacent to an occupied ecosite, which increases the probability that all habitat elements necessary for foraging, mating, nesting, aestivating and hibernating for several generations are included. It is further recommended that a buffer of 90 metres be added to the ELC ecosite polygons to maintain important microhabitat properties (such as substantial leaf litter, decaying logs, and humus-rich soil), to reduce edge effects, and to account for the dispersal of snails into neighbouring habitat. Habitat known to be unsuitable (for example, human-modified landscapes) should be excluded from this buffer.
1.0 Background information
1.1 Species assessment and classification
The following lists provide assessment and classification information for Shagreen (Inflectarius inflectus) and Toothed Globe (Mesodon zaletus). Note: The glossary provides definitions for abbreviations and technical terms in this document.
- SARO List Classification: Endangered
- SARO List History: Endangered (2022)
- COSEWIC Assessment History: Endangered (2019)
- SARA Schedule 1: No schedule, no status
- Conservation Status Rankings: G-rank: G5; N-rank: N1; S-rank: S1
Toothed Globe:
- SARO List Classification: Endangered
- SARO List History: Endangered (2022)
- COSEWIC Assessment History: Endangered (2019)
- SARA Schedule 1: No schedule, no status
- Conservation Status Rankings: G-rank: G5; N-rank: N1?; S-rank: S1?
1.2 Species description and biology
Species descriptions
Shagreen (Inflectarius inflectus (Say, 1821)) belongs to the family Polygyridae, a group of air-breathing land snails. It was first described by Say in 1821 as Helix inflecta, and was subsequently assigned to the genus Triodopsis by Binney in 1878 and Polygyra by Pilsbry in 1900 (Pilsbry 1940). In 1940, Pilsbry again transferred the species to the genus Mesodon, and later the subgenus Inflectarius, which is currently the accepted genus (Turgeon et al. 1998). Shagreen is a distinctive and medium-sized snail (adults measured at maximum shell breadth are 8 – 14 mm) snail with a cream-buff or yellow to brown depressed shell that lacks a hollow in the middle where the whorls come together (Pilsbry 1940). The shell surface has shallow grooves with granules and a scaly outer shell layer (the scales are hair-like in juveniles, but lose this appearance as adults) (COSEWIC 2019a). The shell opening has a lip that is reflected, forming a gutter at its base, and there are three tooth-like denticles visible inside: one long, slightly curved tooth on the upper wall, a tooth at the basal lip, and a blunt, slightly receding tooth at the outer arc of the opening (COSEWIC 2019a) (Figure 1, Left). The body is dark grey and rarely extends out of its shell (Figure 1, Right). Specimens from Middle and Pelee Islands have been sequenced by the Biodiversity Institute of Ontario and their genetic fingerprints are available from the BOLD website (BOLD systems 2022).

Figure 1. Shagreen (Inflectarius inflectus)
Figure 1. Shagreen (Inflectarius inflectus). Photo on left by A. Nicolai (2013); photo on right by Point Pelee National Park Team (2018). The small, closely spaced hatch marks represent millimetres.
Toothed Globe (Mesodon zaletus (Binney, 1837)) also belongs to the family Polygyridae. It was originally described as Helix zaleta by Binney in 1837, but was also known as H. exoleta by Binney in 1851, Mesodon exoleta by Binney in 1878 and Polygyra zaleta by Pilsbry in 1900 (Pilsbry 1940). Pilsbry placed it in its current genus, Mesodon, in 1940. Toothed Globe is a larger snail (adults measured as maximum shell breadth are 2.4 – 3.1 cm) with a yellow, globose-depressed, solid shell (Pilsbry 1940).
The shell has oblique, shallow grooves on its surface, and a reflected, white, nearly three millimetre wide lip in the shell opening of adults (Figure 2). The opening has a white tooth-like denticle on its upper wall. The tissue on the inside of the shell is marked with confluent black spots that are visible through the shell in younger specimens (COSEWIC 2019b). The body is greyish brown or blackish, and paler underneath. No genetic data is available for this species.

Figure 2. Toothed Globe (Mesodon zaletus)
Figure 2. Toothed Globe (Mesodon zaletus), UMMZ105034, recorded by C. Goodrich and M. L. Winslow in 1890 in Leamington, University of Michigan Museum of Zoology, Bryant Walker collection, photo by UMMZ Mollusk Division.
Species biology
Little is known about the biology of either Shagreen or Toothed Globe. Both are air-breathing, terrestrial snails that possess both male and female reproductive organs (simultaneous hermaphrodite) (Pilsbry 1940). Both members of a mating pair may exchange sperm and produce eggs. In most species, snails that are large lay more eggs than smaller ones (Heller 2001) and in temperate regions reproduction usually occurs in spring and late summer with egg clutches deposited in shallow holes excavated in moist soil or litter (Barker 2001). Clutch size for Shagreen and Toothed Globe is unknown (COSEWIC 2019a, 2019b).
During targeted surveys between 2013 and 2018, Shagreen was only observed under logs, not moving, while other snail species (not Mesodon zaletus) found in the same areas were mainly active in the morning hours or after rain (COSEWIC 2019a). The hibernation period for both species is likely from early October until mid-April, depending on environmental conditions each year (COSEWIC 2019a, 2019b). Hibernation sites are unknown for both species, but typical sites in other species are shallow depressions in the forest floor covered with leaf litter or soil at depths of 4 to 10 centimetres (Pearce and Örstan 2006). During prolonged periods of heat and drought in temperate regions, some species of snail go through aestivation, where they remain inactive in moist microhabitats, such as soil, leaf litter and under logs (Nicolai et al. 2011). Other Polygyridae species in aestivation and hibernation with epiphragms (a calcified covering over their shell opening) have been observed in August and November, respectively, during surveys between 2013 and 2018 in Canada (COSEWIC 2019a, 2019b).
Growth typically occurs only during periods of activity (Spring to Fall) (Nicolai 2010; Nicolai et al. 2010). Growth for Shagreen and Toothed Globe are unknown, however, parallels can be made with other snail species of similar sizes. Adult shell size for Shagreen (approximately 10 mm in width) is likely reached after one year with sexual maturity being reached at the same time (COSEWIC 2019a). Shagreen may live two to three years, with an estimated generation time of two years (COSEWIC 2019a). Toothed Globe likely reaches adult shell size (approximately 2 cm in width) after one to two years and sexual maturity after two to three years (COSEWIC 2019b). Adult Toothed Globe can have old, thick and weathered shells (without the coloured outer layer), which indicates that the species is long-lived (i.e., 5 – 10 years), with an estimated generation time of five to six years (COSEWIC 2019b).
Details of diet and feeding behaviour of Shagreen and Toothed Globe are unknown. However, like many other species of litter dwelling snails (COSEWIC 2014a; COSEWIC 2017; COSEWIC 2018), they may eat decaying plants or microfungi on logs on which the snails are found (COSEWIC 2019a). Snails generally play an important role in forest ecosystem functioning through their feeding habits, specifically by aiding in decomposition, nutrient cycling and soil building processes (Mason 1970a, 1970; Jennings and Barkham 1979).
Dispersal and migration strategies for both species are unknown. The daily active movement distances for both species are also unknown, but other members of the family Polygyridae that are similar in size move between 120 and 220 centimetres per day within a home range of 80 to 800 m2 (COSEWIC 2019a, 2019b; Pearce 1990). Shagreen has never been observed actively moving outside a log during recent surveys in Canada and may have a lesser dispersal ability (COSEWIC 2019a). The European species at risk Quimper Snail (Elona quimperiana), living under logs similar to Shagreen, dispersed 500 metres in about 50 years, taking advantage of logs distributed over the site where it was introduced (Lebourcq 2020). The eggs and immature stage of both Shagreen and Toothed Globe were not observed to be dispersed by wind, water, passage by other animals (i.e., through bird intestines, migration), rafting on floating objects or fish, and the likelihood of these methods being used by either species is small (COSEWIC 2019a, 2019b). Due to the poor dispersal capabilities of these species, it is unlikely that dispersal from populations in the United States into Ontario will occur in the near future (COSEWIC 2019a, 2019b). Historical and current habitat loss and degradation are likely factors preventing any northern expansion of the Canadian populations of Shagreen and Toothed Globe (COSEWIC 2019a, 2019b). Furthermore, since neither species is linked to human activities, they are unlikely to be accidentally introduced to new areas, and because neither species feeds on fresh plant material, they are unlikely to be transported via horticultural or agricultural goods (Robinson 1999; Robinson and Slapcinsky 2005).
Predators and parasites of Shagreen and Toothed Globe are not documented. However, it is well documented that trematodes and free swimming or attached flagellates were observed in other Polygyridae (Barger and Hnida 2008; Barger 2011; COSEWIC 2019a; Current 2007). Parasitic mites and nematodes are also commonly observed in snails in general and can cause high mortality, reproductive disturbance, and reduced cold hardiness (Baur and Baur 2005; COSEWIC 2019a; Morand et al. 2004; Örstan 2006, Schüpbach and Baur 2008). Snails are an important food source for a variety of taxa, including salamanders, frogs, toads, turtles, snakes, lizards, birds, shrews, voles, moles, rats, mice, chipmunks, squirrels, sciomyzid fly larvae, firefly larvae, parasitic wasp larvae, beetles, ants, spiders and harvestman (Jordan and Black 2012). Carnivorous gastropods, such as the introduced Draparnaud’s Glass Snail (Oxychilus draparnaudi) and Cellar Glass Snail (Oxychilus cellarius), are predators on other snails and have been observed on Lake Erie islands and the mainland of southwestern Ontario, and may contribute to increased mortality of Shagreen and Toothed Globe (COSEWIC 2019a).
1.3 Distribution, abundance and population trends
Shagreen is distributed across eastern North America, where it is found as far north as southern Ontario, Michigan and New York, east to New York, south to Florida and Texas and as far west as Kansas and Oklahoma (NatureServe 2022). In Canada, Shagreen is historically known from mainland southwestern Ontario (two sites near Leamington) and from five Lake Erie islands. Currently, it is known on two Lake Erie Islands: Middle Island (one subpopulation) and Pelee Island (seven sites comprising nine subpopulations) (Figure 3) (COSEWIC 2019a). The Pelee Island subpopulations are delineated on the basis of a lack of suitable connecting habitat and existing barriers (i.e., roads, unsuitable habitat) that could decrease gene flow, even within sites. While historical records from southwestern Ontario mainland sites and two other islands in Lake Erie exist, it appears to have been extirpated from these areas, as collection efforts between 2013 and 2018 failed to collect any specimens (COSEWIC 2019a). The species may be extant at three historical sites, but these sites have not been resurveyed in the past 20 years.
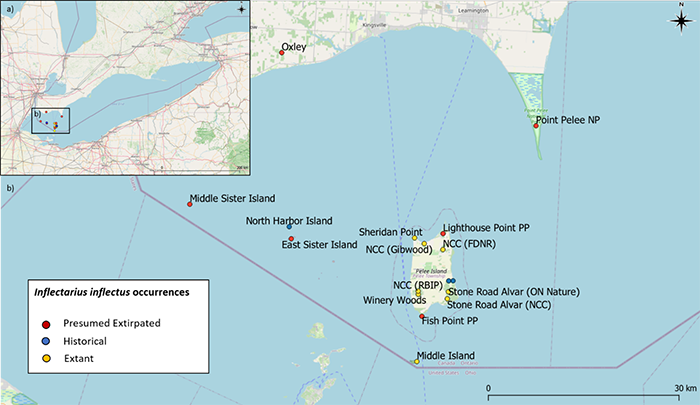
Figure 3. Presumed extirpated, extant and historical distribution of Shagreen in Ontario
Figure 3. Presumed extirpated, extant and historical distribution of Shagreen in Ontario. “Presumed Extirpated” refers to observations older than 20 years which were not observed during recent surveys. “Extant” means live individuals or recently occupied, unweathered shells were found within the last 20 years. “Historical” refers to locations where the species was previously observed, and that provide habitat that is suitable for Shagreen (evaluated from recent photographs, aerial photographs and surrounding visits) but have not been surveyed within the last 20 years (for example, private land).
Generally, clusters of 10 to 20 individuals can be found under a log (COSEWIC 2019a). The most recent record is a cluster of approximately 20 individuals found on Pelee Island in 2020 (observed by Jill Crosthwaite, iNaturalist 2022), indicating that reproduction is occurring. On Middle Island, records from 2013 include three occurrences with 44, 38 and 2 individuals. Seven monitoring plots were established in 2015, randomly distributed over the island. In 2015, four out of these seven plots harboured 1 to 11 individuals each while in 2017 to 2019 only two plots had 1 to 2 individuals each. In 2021, two plots had 3 to 4 individuals each. Given the scarcity of the species, a maximum density of less than 0.1 adults per square metre is expected, the distribution being extremely patchy and linked to log distribution (COSEWIC 2019a). The size of the Canadian population of Shagreen is unknown due to the low collection results and the lack of access to potential habitat on private lands. An estimate of the maximum number of mature individuals within protected areas within Canada is 480,100 (COSEWIC 2019a). This number is based on the assumption that Shagreen density is evenly distributed over the entire protected natural area where it has been recorded over the last 20 years on Pelee and Middle Islands. Due to its patchy, clumped distribution over this area, this number should be viewed with caution (COSEWIC 2019a).
Toothed Globe shares a similar distribution to Shagreen across North America, with a northern limit in southern Ontario, Michigan and New York, east to New York, as far south as South Carolina across to Texas (excluding Florida) and west to Oklahoma, Missouri and Iowa (COSEWIC 2019b; NatureServe 2022). In Canada, Toothed Globe is historically known from several islands in Lake Erie and from Essex and Middlesex counties on the mainland of southwestern Ontario (Figure 4). It may still occur on private and First Nations land in Essex and Middlesex counties that have not been surveyed and where intact habitat remains, as well as any other unsurveyed accessible sites within the historically known range (COSEWIC 2019b). Recent search efforts from 2013 to 2017 failed to record living specimens in accessible areas. It appears to have been extirpated from most known southwestern Ontario sites, including the Lake Erie islands (COSEWIC 2019b). In 2013, two weathered (without the outer coloured layer), empty snail shells were found on two of the Lake Erie islands: one under a large pile of old shells and one under a 50 centimetre thick mulch layer with many other shells. The conditions in which the two sole shells of this species were found indicate that the species is probably extirpated from these islands (COSEWIC 2019b). Population trends and estimates are not available for Toothed Globe since no living individuals have been collected since 1994.
The current distribution is based on large historical surveys conducted by John Oughton (1930-1940) (Oughton 1948), by Wayne Grimm (1970-1995) (Grimm 1996), by Michael Oldham (1992-2012) (all his 2,349 georeferenced records can be consulted through NHIC; see also COSEWIC 2019), by Jane Bowles (1994) (113 georeferenced records analyzed by A. Nicolai and R. Forsyth) and by A. Nicolai in 2012 (364 georeferenced records). Recent search effort since 2013 resulted in 137 surveyed sites with 585 person-hours. As suitable habitat decreased considerably during the last century in the very densely populated most western part of Southwestern Ontario (most known occurrence sites of the two species are in this area) the probability that new occurrence sites will be discovered is low. Nevertheless, some private land (for example, White Oak Woods, but also other still unknown sites) and First Nation land (especially the historically known occurrence site for Toothed Globe) may still harbour old growth forest with low human impact that would be suitable habitat for the two species. In the more eastern part of Southwestern Ontario (between east of London to the historical Hamilton occurrence site of Toothed Globe), many more publicly accessible sites can be surveyed on the Niagara Escarpment, although historical surveys have no records from this area.
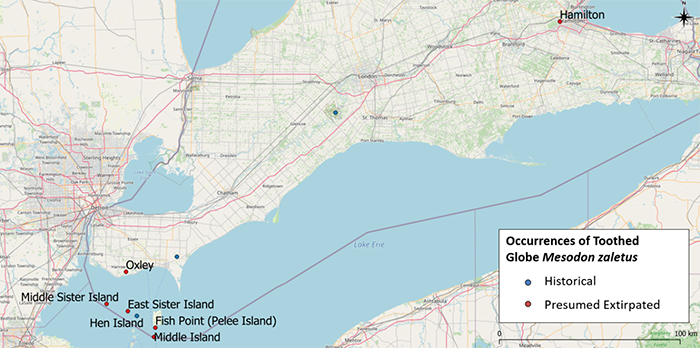
Figure 4. Presumed extirpated and historical distribution of Toothed Globe in Ontario
Figure 4. Presumed extirpated and historical distribution of Toothed Globe in Ontario. Presumed extirpated
refers to observations older than 20 years which were not observed during recent surveys. Historical
refers to locations where the species was previously observed, and that provide habitat that is suitable for Toothed Globe (evaluated from recent photographs, aerial photographs and surrounding visits) but have not been surveyed within the last 20 years (for example, private land).
Some habitat in most historically occupied sites of the two species is still intact, and the exact reasons for the presumed extirpations are unknown. Habitat on the Lake Erie islands was altered by Double-crested Cormorant nesting and heavy storms flooding the islands. Currently these islands have less suitable habitat.
1.4 Habitat needs
According to Hubricht (1985), Shagreen may be found in a variety of habitats under anything from natural objects (logs, rocks and leaf litter in woods) to human-made items (old ties along railroads and roadsides). In Canada, Shagreen lives in rocky or open woods (including Common Hackberry (Celtis occidentalis) woods), open deciduous woods and a wooded alvar with Chinquapin Oak (Quercus muehlenbergii) and is always found under logs (COSEWIC 2019a). The species’ occurrences can be overlayed on Ecological Land Classification (Lee et al. 1998) maps showing an affinity for particular habitat types on Pelee Island: Chinquapin Oak – Nodding Onion Treed Alvar; Fresh-Moist Sugar Maple – Lowland Deciduous Forest; Dry-Fresh Oak – Maple – Hickory Deciduous Forest; Dry-Fresh Calcareous Bedrock Deciduous Woodland; and Hackberry Calcareous Treed Rock Barren. On Middle Island it is found all over the island in various types of Hackberry Forest following the vegetation community analysis of North - South Environmental Inc. (2004). According to COSEWIC (2019a), all currently known Shagreen sites are surrounded by unsuitable arable land or water.
Toothed Globe was historically known to occupy similar habitat in Canada as it does in the United States, where it is most common on river bluffs, but may also be found in ravines and on mountainsides up to about 1,500 metres in elevation (COSEWIC 2019b; Hubricht 1985). It prefers slopes with thick leaf litter over a rich humus (Pilsbry 1940) in cool hardwood or mixed forests, especially on steep slopes along rivers (Hotopp and Winslow 2012). Caldwell et al. (2014) found that Toothed Globe was more abundant in Red Oak dominated forests than in other habitat types on Magazine Mountain (Arkansas), while Coney et al. (1982) found the snails in limestone areas of old growth forests. Toothed Globe may be found in both acidic and neutral/calcareous habitats (Nekola 2010).
General microhabitat requirements for terrestrial gastropods are moist, temperature-buffered refuges that provide shelter against extreme weather (Barker 2001). These refuges are closely linked to food availability, such as decaying plant material, but also fungi hyphae in decaying leaf litter and humus for Toothed Globe (Pilsbry 1940), and probably in decaying logs for Shagreen (COSEWIC 2019a). It is widely understood that terrestrial gastropods seek different microhabitat conditions to support varying biological functions such as foraging, mating, nesting, aestivating and hibernating, and the availability of microhabitat may be the most important factor limiting terrestrial snail abundance (Burch and Pearce 1990). Further research is required in this area specific to Shagreen and Toothed Globe.
1.5 Limiting factors
Shagreen and Toothed Globe are both near the northern limits of their distribution and further expansion north is likely limited by harsh winters, human-caused habitat fragmentation and loss (Gibson et al. 2009), and physical barriers, such as large bodies of water (COSEWIC 2019a, 2019b). Other limiting factors that may restrict gene flow among subpopulations for both species include low dispersal ability and low physiological resistance to a fluctuating environment (for example, temperature and humidity) (COSEWIC 2019a, 2019b). Availability of moist refuges, such as under logs, that buffer environmental fluctuations is likely a limiting factor for population growth at the microhabitat scale (Burch and Pearce 1990).
1.6 Threats to survival and recovery
A threat assessment for Shagreen was compiled for its COSEWIC report (2019a) and included information from extant subpopulations on Middle and Pelee Islands and sites with potential habitat where empty shells were observed between 2013 and 2018 (mainland subpopulations and other Lake Erie islands where the species is considered extirpated were not included). Since Toothed Globe has not been observed in Canada for some time, it is uncertain if it is still extant here, thus a threats assessment was not completed during the preparation of its COSEWIC report (2019b). It is very likely facing similar threats to Shagreen based on the current expected limited range of both species. Therefore, the following threats can be assumed for both species.
Invasive/hyperabundant species and resulting habitat alterations
Since the early 1980’s, Lake Erie islands, especially Middle Sister, East Sister and Middle Islands, have seen a dramatic increase in Double-crested Cormorant (Phalacrocorax auritus) nesting colonies (COSEWIC 2017). The guano of these birds leads to soil chemistry modification, tree dieback, canopy cover loss, reduced plant species richness and an increased proportion of exotic species (Boutin et al. 2011; Rush et al. 2013; Hebert et al. 2014). They may be the primary reason why Shagreen (East and Middle Sister Islands) and Toothed Globe (East Sister, Middle and Middle Sister Islands) have been extirpated from these islands, as they have been identified as a threat to Eastern Banded Tigersnail (Anguispira kochi kochi) for the reasons mentioned above (COSEWIC 2019a, 2019b). Parks Canada has been culling Double-crested Cormorants since 2008 on Middle Island (Thorndyke and Dobbie 2013) and vegetation has recovered, although how this will affect snail populations, if at all, is unknown.
Several highly invasive plant species in southern Ontario, including Garlic Mustard (Alliaria petiolata), are found on Pelee and Middle Islands. Garlic Mustard is known to displace native vegetation and alter soil nutrient cycles, which slows restoration of native plant species such as spring ephemeral wildflowers (Catling et al. 2015). Japanese Chaff Flower (Achyranthes japonica) has recently been detected on Middle and East Sister Islands (Dobbie pers. comm. 2022). This plant outcompetes native species and forms large, dense stands in floodplains, forested wetlands, shorelines and other disturbed edge habitat (Michigan Invasive Species 2022). The effect of these invasive plant species on snail populations is not yet known. In some cases, invasive plants can lead to a decrease in endangered snail abundance (Stoll et al. 2012), but can also have a positive impact on snail diversity (Utz et al. 2018).
Introduced non-native earthworms have recently become established in Canada and have altered forest floor habitats by reducing or eliminating the natural leaf litter layer and digging up and mixing the mineral soil with the organic surface layer (CABI 2016). Through these habitat alterations, invasive earthworms may indirectly alter terrestrial snail communities (Forsyth et al. 2016). Invasive earthworms are present on the north shore of Lake Erie (Evers et al. 2012), Pelee Island (Reynolds 2011) and elsewhere in Ontario (Reynolds 2014). Earthworms in the genus Amynthas have been introduced from Asia to Essex County (Reynolds 2014), and are known to reduce surface leaf litter very quickly where snails live (Qiu and Turner 2017).
Exotic terrestrial gastropods are also a potential threat (Whitson 2005; Grimm et al. 2010) for Shagreen and Toothed Globe through aggression (Kimura and Chiba 2010), density effects, food competition (Baur and Baur 1990) and competition for shelter (COSEWIC 2019a). Several species of exotic gastropods are widespread in southern Ontario, including Dusky Arion (Arion fuscus), Grey Field Slug (Deroceras reticulatum) and Grove Snail (Cepaea nemoralis), and specifically on Lake Erie islands (the carnivorous Draparnaud’s Glass Snail and Cellar Glass Snail), and may directly affect Shagreen and Toothed Globe (COSEWIC 2019a, 2019b; Mahlfeld 2000). Leopard Slug (Limax maximus) has recently been recorded on Middle Island (Nicolai pers. obs.) and may also have an effect on Shagreen and Toothed Globe populations due to competition for food.
Birds introduced to some parts of Ontario for recreational hunting, such as Wild Turkeys (Meleagris gallopavo) and Ring-necked Pheasants (Phasianus colchicus) may pose a threat to Shagreen and Toothed Globe because both species are omnivorous and feed on snails (Sandilands 2005). These bird species have recently been identified as ongoing threats to other endangered snail species (COSEWIC 2014b; 2017; 2018). Their impacts on Shagreen and Toothed Globe are unknown, but may be reduced as both snail species hide under logs and rock piles, making them less accessible to birds (COSEWIC 2019a, 2019b).
Parasites, like mites and nematodes, may become a threat to the species if the parasites’ population dynamics change due to ongoing ecological changes. Haeussler et al. (2012) found that longer hibernation periods in the Copse Snail (Arianta arbustorum), decreased parasitic mite infections, whereas a shortened hibernation period found higher mite infections which decreased winter survival of host snails. Further study on the effects of mite infection in Shagreen and Toothed Globe must be completed, but increased infestation may lead to higher mortality in subpopulations.
Climate change and severe weather
Shagreen and Toothed Globe are considered highly vulnerable to the effects of climate change (unseasonable spring frosts, absence of snow cover, droughts, etc.) because they are sensitive to specific microhabitat conditions and they have a low adaptive capacity (limited physiological plasticity and low intrinsic and extrinsic dispersal possibilities because they live on islands) (COSEWIC 2019a, 2019b). Climate change models suggest that southwestern Ontario will experience more extreme weather events including droughts, floods and temperature extremes (Varrin et al. 2007). Snails may be vulnerable to increasing average temperatures accompanied by increased incidences of drought (Pearce and Paustian 2013). Numbers of total snails found during surveys in southern Ontario in 2016, considered a dry year, were low compared to 2015, which indicates some vulnerability to drought (COSEWIC 2019b). Spring frost is more frequent with increasing average temperatures (Augspurger 2013), which can cause spring mortality in snails when snow cover is absent (COSEWIC 2019a). Shagreen is a medium-sized snail, which is less susceptible to freezing than larger snails, such as Toothed Globe, but both species still need snow cover and temperature-buffered microhabitats to shelter in winter (Ansart et al. 2014).
Storms are also a major natural disturbance on Middle Island (Parks Canada 2008). The south side of the island is particularly susceptible to severe storms, and large piles of weathered shells of several species of snails were observed during the 2013–2018 surveys, perhaps indicating massive mortality due to these storms (COSEWIC 2019a). Flooding from violent storms occasionally immerses the south side of the island, and may also affect snail populations and their ability to recolonize other Lake Erie islands (COSEWIC 2019a).
Prescribed fire
Prescribed burns are an important management tool for prairie and forest conservation (Williams 2000) and are used to limit the invasion of exotic species (Brooks and Lusk 2008) and to promote growth and reproduction of native prairie species (Towne and Owensby 1984). Burning directly and indirectly affects survival of ground dwelling animals, including snails (Nekola 2002), by reducing and modifying organic substrates used as shelters, increasing soil evaporation and destroying the upper part of the soil and leaf litter habitat, which are important for the survival of litter-soil organisms (Bellido 1987; Knapp et al. 2009). Portions of the Stone Road Alvar on Pelee Island were subjected to prescribed burns by Ontario Nature and Essex Regional Conservation Authority in 1993, 1997, 1999 and 2005 (NCC 2008), as well as in 2019 accompanied by an impact study implemented by Ontario Nature, including snail monitoring done by A. Nicolai. Although no live snails or shells of Shagreen have been found in the Stone Road Alvar, the threat from fire should be considered. Direct impacts from fire on snail populations are reduced when available habitat is widespread and recolonization from nearby areas is possible. However, when habitat areas are small, large fires are considered detrimental to subpopulations (COSEWIC 2019a). Small, patchy fires that are restricted to a smaller area would be less harmful (COSEWIC 2019a).
Pollution
Heavy metals and road salt in close proximity to roads are a threat to land snails (Viard et al. 2004) because they decrease food consumption, growth and fecundity (Laskowski and Hopkin 1996) as a result of accumulation in the soil and food plants (Notten et al. 2005).
The effects of pesticides on terrestrial gastropods are poorly known. Laboratory studies have shown that some herbicides increase mortality of aquatic snails that are infected with parasites (Koprivnikar and Walker 2011) and could affect reproduction in terrestrial snails (Druart et al. 2011), while other studies have found that terrestrial snails were not impacted by herbicides in agricultural (Roy et al. 2003) or forested (Hawkins et al. 1997) landscapes. The increasingly used neonicotinoid insecticides were found not to be harmful to Grey Fieldslug but did have negative effects on mollusk-predating arthropods (Douglas et al. 2015). The discovery of exotic, invasive Japanese Chaff Flower (Achyranthes japonica) in 2018 on Middle Island and East Sister Islands has prompted Parks Canada and Ontario Parks to attempt to eradicate it through hand pulling of small plants and spot application of Glyphosate to larger plants or colonies (Dobbie pers. comm. 2022). Glyphosate is a commonly used herbicide and has been found to have no effect on mortality or growth in the terrestrial Garden Snail (Helix aspersa) (Druart et al. 2011), but in high concentrations can have an effect on survival and development, as evidenced in the aquatic Golden Apple Snail (Pomacea canaliculate) (Xu et al. 2017). The effects of pesticides on Shagreen and Toothed Globe are unknown, however the close proximity of agricultural land to wooded areas in southwestern Ontario may expose snails to pesticide drift (COSEWIC 2019b).
Human disturbance
Southwestern Ontario is densely populated. Most of the land has been transformed to farmland or is urbanized. Very few natural sites of more than a few hectares are remaining. Accessible Lake Erie islands where the species occur have no human activity except research visits in the summer. Private islands have some summer visits (North Harbour) or fishing activity (Hen Island). On Pelee Island, the major human activity is farming (soy beans and grape vines). On the mainland, occurrence sites are surrounded by farmland that mainly produces vegetables and soy beans.
Gastropod populations may be fragmented by paved roads or tracks as narrow as three metres (Wirth et al. 1999) because snails tend not to cross roads (Baur and Baur 1990). Reck and van der Reer (2015) present a study of local extinctions in a population of Rotund Disc (Discus rotundatus) in Germany after its original habitat became unsuitable due to roads disconnecting habitat.
Road mortality has been recognized as a threat for wildlife in protected areas, such as Point Pelee National Park (Parks Canada 2007). Since Shagreen rarely moves away from under logs, they are not likely to be affected by passing traffic (COSEWIC 2019a, 2019b). It is unknown how road mortality may affect Toothed Globe, but impacts would likely be minimal. Tourism on Pelee Island is increasing and Stone Road Alvar is a popular destination for ecotourism with increasing traffic on the road crossing the Alvar site.
Trampling is a negligible threat to these species because they live under logs and rocks, but displacement of these habitats may alter the microhabitat conditions (COSEWIC 2019a). While there is a short loop trail, large parts of Stone Road Alvar are not accessible due to high vegetation density and absence of trails, and Middle Island is closed from March 1 to September 1 annually to protect water bird colonies during nesting season (COSEWIC 2019a).
1.7 Knowledge gaps
The current distribution of Toothed Globe is unknown. There have been no documented living specimens of Toothed Globe in Ontario since 1994 but it is possible it has been overlooked (COSEWIC 2019b). Much of the full historic area of occupancy in Ontario of both Shagreen and Toothed Globe was surveyed from 2013 to 2018, but some sites on private property and First Nations lands were not accessed leaving the potential for these species to have small subpopulations remaining (COSEWIC 2019a, 2019b). It is unknown if they still persist in other recently unsurveyed sites within the historically known range and in other sites with habitat types where historical surveys were less common.
Because current distribution data are either limited (Shagreen) or unavailable (Toothed Globe), population trends in Ontario are unknown, and threats to any extant subpopulations are either site-specific (for example, Double-crested Cormorant nesting) or global (for example, climate change).
The direct causes for the historical decline of these species is habitat loss or degradation. Remaining subpopulations occur in protected areas and direct habitat loss is less likely in the near future, but remains an ongoing threat. The likelihood of ongoing decline is difficult to predict because of the limited biological knowledge available for each species. Basic biological knowledge, such as diet, reproduction, predators/parasites and habitat requirements, as well as dispersal strategies and the impact of invasive species, would provide better insight into the factors that are most important for the survival or decline of these species, and would provide important insights into recovery viability.
1.8 Recovery actions completed or underway
To date, no specific recovery actions have been implemented for Toothed Globe.
A study of prescribed burn impacts on species at risk on Stone Road Alvar implemented by Ontario Nature will include targeted Shagreen surveys in 2022 and 2023. The same study included snail surveys from 2017 to 2020 implemented by A. Nicolai, but Shagreen was not specifically targeted, nor was it found in the burn area. Shagreen occurs mainly on Nature Conservancy Canada land on Pelee Island where snail-focused habitat enhancement, public outreach for awareness and surveys are conducted by trained staff (Dobbie pers. comm. 2022). On Middle Island, Parks Canada is implementing snail species at risk monitoring twice a year including Shagreen since 2015 (Dobbie pers. comm. 2022). The current design of the monitoring allows it to detect the species, follow abundance in a semi-quantitative way over time, verify that reproduction occurs and describe gastropod community composition.
2.0 Recovery
2.1 Recommended recovery goal
The recommended recovery goal for Shagreen is to maintain, and, where possible, support the natural expansion of the current subpopulations. The recommended recovery goal for Toothed Globe is to increase knowledge of the species and its habitat, and, if populations are found to exist, maintain and support the natural expansion of the subpopulations.
2.2 Recommended protection and recovery objectives
The recovery goals for these species are focused on addressing knowledge gaps, mitigating threats and enhancing habitat to allow for long-term population persistence and expansion in Ontario. To achieve these goals, recommended short-term recovery objectives are identified below.
- Engage government land managers, private landowners, and Indigenous communities in surveying suitable habitats to determine the current distribution of Shagreen in Ontario and whether Toothed Globe is still extant in the province
- Assess and mitigate threats to all extant subpopulations, and, where appropriate, historical/presumed extirpated occurrence sites
- Conduct and/or support research that fills knowledge gaps related to biology, population size, and habitat requirements that inform recovery efforts
- Enhance and/or create habitat, where feasible and necessary, to increase habitat availability for extant subpopulations
2.3 Recommended approaches to recovery
| Relative priority | Relative timeframe | Recovery theme | Approach to recovery | Threats or knowledge gaps addressed |
|---|---|---|---|---|
| Critical | Short-term | Communication, Education and Outreach | 1.1 Develop identification material to aid in accurate recognition of Shagreen and Toothed Globe, including how to distinguish them from other similar species
|
Knowledge gaps:
|
| Critical | Short-term | Communication, Education and Outreach | 1.2 Engage landowners, land managers, Indigenous communities, non-governmental organizations and volunteers (e.g., local naturalists, land stewards, experts) to undertake surveys in the search for these snails to determine presence or absence at historical/presumed extirpated sites and potential new sites that have not been surveyed yet
|
Knowledge gaps:
|
| Critical | Short-term | Communication, Education and Outreach | 1.3 Encourage the recording, sharing and transfer of Traditional Ecological Knowledge, where appropriate, to increase knowledge of the species and support future recovery efforts
|
Knowledge gaps:
|
| Critical | Short-term | Communication, Education and Outreach | 1.4 Develop standardized survey protocols for the two species and monitoring protocols for Shagreen
|
Knowledge gaps:
|
| Relative priority | Relative timeframe | Recovery theme | Approach to recovery | Threats or knowledge gaps addressed |
|---|---|---|---|---|
| Critical | Short-term | Management | 2.1 Develop and implement site-specific management plans that identify and mitigate threats to snails and their habitat
|
Threats:
|
| Critical | Short-term | Management | 2.2 Conduct research in the effects of Double-crested Cormorant colonies on snails
|
Threats:
Knowledge gaps:
|
| Critical | Short-term | Management | 2.3 Conduct research on the implications of invasive species that may threaten snails and/or their habitat, including:
|
Threats:
Knowledge gaps:
|
| Necessary | Long-term | Research | 2.4 Conduct research on the implications of climate change and severe weather on snails and their habitat
|
Threats:
Knowledge gaps:
|
| Necessary | Long-term | Inventory, monitoring and assessment | 2.5 Assess the impact of habitat management actions on the two species
|
Threats:
Knowledge gaps:
|
| Beneficial | Long-term | Inventory, monitoring and assessment | 2.6 Identify extant or historical/presumed extirpated habitat that is more vulnerable to threats from flooding, erosion, fire and development
|
Threats:
Knowledge gaps:
|
.
| Relative priority | Relative timeframe | Recovery theme | Approach to recovery | Threats or knowledge gaps addressed |
|---|---|---|---|---|
| Critical | Short-term | Research | 3.1 Engage the academic community to participate in researching knowledge gaps such as:
|
Threats:
Knowledge gaps:
|
| Critical | Short-term | Research, Inventory, Monitoring and Assessment | 3.2 Monitor activity to determine population dynamics, home range size, dispersal ability and patterns of habitat occupancy
|
Knowledge gaps:
|
| Necessary | Long-term | Management, Protection | 3.3 Investigate feasibility and success of population augmentation measures
|
Threats:
|
| Relative priority | Relative timeframe | Recovery theme | Approach to recovery | Threats or knowledge gaps addressed |
|---|---|---|---|---|
| Critical | Ongoing | Management, Protection, Stewardship | 4.1 Identify, protect and/or create refuge areas for snails to move into in times of extreme temperatures and/or droughts
|
Threats:
|
| Beneficial | Long-term | Management, Protection | 4.2 Identify habitat restoration opportunities that encourage connectivity between habitats to allow dispersal
|
Threats:
|
| Beneficial | Long-term | Monitoring and Assessment | 4.3 Monitor and evaluate habitat enhancement/ creation activities and adjust actions if needed | Threats:
|
2.4 Area for consideration in developing a habitat regulation
Under the ESA, a recovery strategy must include a recommendation to the Minister of the Environment, Conservation and Parks on the area that should be considered if a habitat regulation is developed. A habitat regulation is a legal instrument that prescribes an area that will be protected as the habitat of the species. The recommendation provided below by the author will be one of many sources considered by the Minister, including information that may become newly available following the completion of the recovery strategy should a habitat regulation be developed for this species.
Information on the spatial limits of habitat used and dispersal by Shagreen and Toothed Globe is lacking. When information on home range size, dispersal ability and key habitat features critical for supporting the species’ lifecycle becomes available, the area prescribed as habitat could be described more precisely and should be revisited. Based on the best information available, it is recommended that all Ecological Land Classification (ELC) ecosites (Lee et al. 1998) occupied by an extant subpopulation be prescribed as habitat in a habitat regulation. The entire ecosite is recommended because given the rarity of these species, they may be present throughout the ecosite but not detected everywhere. Additionally, like other snail species, they may use different habitat patches within different ecosites in different seasons for various biological functions such as feeding and aestivation/hibernation (Burch and Pearce 1990). Therefore, it is recommended that the regulated area should be defined using a contiguous ecological area encompassing all occupied ecosites and any suitable unoccupied ecosites immediately adjacent to an occupied ecosite (Figure 5). This recommendation increases the probability that all habitat elements necessary for foraging, mating, nesting, aestivating and hibernating for several generations are included, supports long-term recovery and helps protect against false-absence data for these cryptic species. Ecosites currently believed to be suitable are described in section 1.4 of this recovery strategy, but this list may not be exhaustive as further research is required.
It is further recommended for the two species that a buffer of 90 metres be added to the defined ELC ecosite polygons (inclusive of both occupied ecosites and adjacent suitable unoccupied ecosites). This buffer area is necessary to maintain important microhabitat properties and to reduce edge effects (Harper et al. 2005). The buffer may include habitat unsuitable for long-term occupancy, but should exclude habitat known to be unsuitable for maintaining microhabitat, such as human-modified landscapes, existing infrastructure and waterbodies (for example, roads, farmland, urban areas, gardens, parks on the mainland and lakes). While the primary intent of the 90-metre buffer is to maintain suitable microhabitat, this buffer also accounts for temporary use of unsuitable neighbouring habitat based on the longest short-term dispersal distance measured in Polygyridae (32 m) (Edworthy et al. 2012).
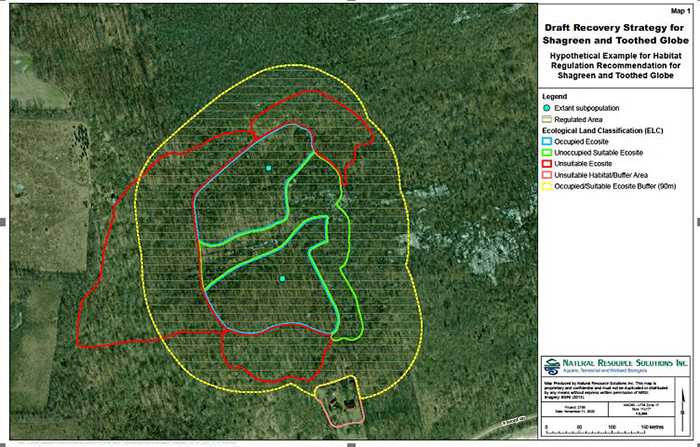
Figure 5. Schematic application of the habitat regulation recommendation for Shagreen and Toothed Globe
Glossary
- Aestivation
- Prolonged dormancy during a hot or dry period
- Committee on the Status of Endangered Wildlife in Canada (COSEWIC)
- The committee established under section 14 of the Species at Risk Act that is responsible for assessing and classifying species at risk in Canada
- Conservation status rank
- A rank assigned to a species or ecological community that primarily conveys the degree of rarity of the species or community at the global (G), national (N) or subnational (S) level. These ranks, termed G-rank, N-rank and S-rank, are not legal designations. Ranks are determined by NatureServe and, in the case of Ontario’s S-rank, by Ontario’s Natural Heritage Information Centre. The conservation status of a species or ecosystem is designated by a number from 1 to 5, preceded by the letter G, N or S reflecting the appropriate geographic scale of the assessment. A question mark ‘?’ denotes an inexact numeric rank. The numbers mean the following:
- 1 = critically imperiled
- 2 = imperiled
- 3 = vulnerable
- 4 = apparently secure
- 5 = secure
- NR = not yet ranked
- Denticle
- A small tooth or tooth-like projection
- Endangered Species Act, 2007 (ESA)
- The provincial legislation that provides protection to species at risk in Ontario
- Globose
- Having the form of a globe; spherical
- Species at Risk Act (SARA)
- The federal legislation that provides protection to species at risk in Canada. This Act establishes Schedule 1 as the legal list of wildlife species at risk. Schedules 2 and 3 contain lists of species that at the time the Act came into force needed to be reassessed. After species on Schedule 2 and 3 are reassessed and found to be at risk, they undergo the SARA listing process to be included in Schedule 1.
- Species at Risk in Ontario (SARO) List
- The regulation made under section 7 of the Endangered Species Act, 2007 that provides the official status classification of species at risk in Ontario. This list was first published in 2004 as a policy and became a regulation in 2008 (Ontario Regulation 230/08).
- Pesticide drift
- The movement of pesticide dust or droplets through the air at the time of application or soon after to any site other than the area intended
- Whorl
- A pattern of spirals or concentric circles
List of abbreviations
- BOLDsystems
- Barcode of Life Data System
- CLEF
- Conference and Labs of the Evaluation Forum
- COSEWIC
- Committee on the Status of Endangered Wildlife in Canada
- ESA
- Ontario’s Endangered Species Act, 2007
- ISBN
- International Standard Book Number
- SARA
- Canada’s Species at Risk Act
- SARO List
- Species at Risk in Ontario List
- UMMZ
- University of Michigan Museum of Zoology
References
Ansart, A., A. Guiller, O. Moine, M.C. Martin, and L. Madec. 2014. Is cold hardiness size-constrained? A comparative approach in land snails. Evolutionary Ecology 28:471–493.
Augspurger, C.K. 2013. Reconstructing patterns of temperature, phenology, and frost damage over 124 years: spring damage risk is increasing. Ecology 94:41–50.
Barger, M.A. 2011. Tests of ecological equivalence of two species of terrestrial gastropods as second intermediate hosts of Panopistus pricei (Trematoda: Brachylaimidae). Journal of Parasitology 97:8–13.
Barger, M.A. and J.A. Hnida. 2008. Survey of trematodes from terrestrial gastropods and small mammals in southeastern Nebraska, USA. Comparative Parasitology 75:308–314.
Barker, G.M. 2001. The Biology of Terrestrial Molluscs. CABI Publishing, New York, New York. 558 pp.
Baur, A. and B. Baur. 2005. Interpopulation variation in the prevalence and intensity of parasitic mite infection in the land snail Arianta arbustorum. Invertebrate Biology 124:194–201.
Baur, B. and A. Baur. 1990. Experimental evidence for intra- and interspecific competition in two species of rock-dwelling land snails. Journal of Animal Ecology 59:301–315.
Bellido, A. 1987. Field Experiment about direct effect of a heathland prescribed fire on microarthropod community. Revue d’Ecologie et de Biologie du Sol 24:603–633.
BOLDsystems (Barcode of Life Data System). 2022. [Accessed April 2022].
Brooks, M. and M. Lusk. 2008. Fire Management and Invasive Plants: a Handbook. United States Fish and Wildlife Service, Arlington, Virginia, 27 pp.
Boutin, C., T. Dobbie, D. Carpenter, and C.E. Hebert. 2011. Effects of double-crested cormorants (Phalacrocorax auritus Less.) on island vegetation, seedbank, and soil chemistry: evaluating island restoration potential. Restoration Ecology 19:720–727.
Burch, J.B. and T.A. Pearce. 1990. Terrestrial gastropods. Pp. 201–309. In: D.L. Dindal (ed.). Soil Biology Guide. John Wiley and Sons, New York, New York.
CABI (CAB International). 2016. Invasive Species Compendium. Datasheet Lumbricus rubellus. [Accessed April 2022].
Caldwell, R.S., J.E. Copeland, G.L. Mears, and D.A. Douglas. 2014. Notes on the natural history and ecology of Inflectarius magazinensis (Pilsbry and Ferriss, 1907) (Gastropoda: Polygyridae), the Magazine Mountain Shagreen. American Malacological Bulletin 32:211–216.
Catling, P.M., G. Mitrow, and A. Ward. 2015. Major invasive alien plants of natural habitats in Canada. 12. Garlic Mustard, Alliaire officinale: Alliaria petiolata (M. Bieberstein) Cavara & Grande. CBA/ABC Bulletin 48(2):51–60.
Coney, C.C., W.A. Tarpley, and R. Bohannan. 1982. Ecological studies of land snails in the Hiawassee River Basin of Tennessee, U.S.A. Malacological Review 15:69–106.
COSEWIC. 2014a. Conservation Prioritization of Ontario and Quebec Terrestrial Molluscs. A COSEWIC Special Project Report. Committee on the Status of Endangered Wildlife in Canada. Ottawa. 229 pp.
COSEWIC. 2014b. COSEWIC assessment and status report on the Broad-banded Forestsnail Allogona profunda in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. xi + 53 pp
COSEWIC. 2017. COSEWIC assessment and status report on the Eastern Banded Tigersnail Anguispira kochi kochi and the Western Banded Tigersnail Anguispira kochi occidentalis in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. x + 82 pp.
COSEWIC. 2018. COSEWIC assessment and status report on Striped Whitelip Webbhelix multilineata in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. x + 62 pp.
COSEWIC. 2019a. COSEWIC assessment and status report on the Shagreen Inflectarius inflectus in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. x + 51 pp.
COSEWIC. 2019b. COSEWIC assessment and status report on the Toothed Globe Mesodon zaletus in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. x + 38 pp.
Current, W.L. 2007. Cryptobia sp. in the snail Triodopsis multilineata (Say): fine structure of attached flagellates and their mode of attachment to the spermatheca. Journal of Eukaryotic Microbiology 27:278–287.
Douglas, M.R., and J.F. Tooker. 2015. Large-scale deployment of seed treatments has driven rapid increase in use of neonicotinoid insecticides and preemptive pest management in U.S. field crops. Environmental Science and Technology 49:5088–5097.
Druart, C.M., M. Millet, R. Scheifler, O. Delhomme, and A. de Vaufleury. 2011. Glyphosate and glufosinate-based herbicides: fate in soil, transfer to, and effects on land snails. Journal of Soil Sediments 11:1373–1384.
Edworthy, A.B., K.M.M. Steensma, H.M. Zandberg, and P.L. Lilley. 2012. Dispersal, home-range size, and habitat use of an endangered land snail, the Oregon forestsnail (Allogona townsendiana). Canadian Journal of Zoology 90(7):875–884.
Evers, A.K., A.M. Gordon, P.A. Gray, and W.I. Dunlop. 2012. Implications of a potential range expansion of invasive earthworms in Ontario’s forested ecosystems: a preliminary vulnerability analysis. Climate Change Research Report CCRR-23. Science and Information Resources Division. Ontario Ministry of Natural Resources, Ottawa, Ontario. 46 pp.
Forsyth, R.G., P. Catling, B. Kostiuk, S. McKay-Kuja, and A. Kuja. 2016. Pre-settlement snail fauna on the Sandbanks Baymouth Bar, Lake Ontario, compared with nearby contemporary faunas. Canadian Field-Naturalist 130:152–157.
Gibson, S.Y., R.C. Van der Marel, and B.M. Starzomski. 2009. Climate change and conservation of leading-edge peripheral populations. Conservation Biology 23:1369–1373.
Grimm, F.W. 1996. Terrestrial molluscs. In I.M. Smith, Assessment of species diversity in the Mixedwood Plains ecosystem. Ecological Monitoring and Assessment Network. Canada. CD ROM.
Grimm, F.W., R.G. Forsyth, F.W. Schueler, and A. Karstad. 2010. Identifying Land Snails and Slugs in Canada: Introduced Species and Native Genera. Canadian Food Inspection Agency, Ottawa, Ontario. 168 pp.
Haeussler, E.M., J. Pizá, D. Schmera, and B. Baur. 2012. Intensity of parasitic mite infection decreases with hibernation duration of the host snail. Parasitology 139(8):1038–1044.
Harper, K.A., S.E. MacDonald, P.J. Burton, J. Chen, K.D. Brosofske, S.C. Daunders, E.S. Euskirchen, D. Roberts, M.S. Jaiteh, P. Esseen. 2005. Edge Influence on Forest Structure and Composition in Fragmented Landscapes. Conservation Biology 19(3):768-782.
Hawkins, J.W., M.W. Lankester, R.A. Lautenschlager, and F.W. Bell. 1997. Effects of alternative conifer release treatments on terrestrial gastropods in northwestern Ontario. The Forestry Chronicle 73(1):91–98.
Hebert, C.E., J. Pasher, D.V.C. Weseloh, T. Dobbie, S. Dobbyn, D. Moore, V. Minelga, and J. Duffe. 2014. Nesting Cormorants and Temporal Changes in Island Habitat. The Journal of Wildlife Management 78(2):307–313.
Heller, J. 2001. Life history strategies. Pp. 413–445. In: G.M. Barker (ed.). The Biology of Terrestrial Molluscs. CABI Publishing, New York, New York.
Hotopp, K. and M. Winslow. 2012. Virginia land snails: Mesodon zaletus (A. Binney, 1837). Carnegie Museum of Natural History. Website: [Accessed April 2022].
Hubricht, L. 1985. The distributions of the native land mollusks of the Eastern United States. Fieldiana Zoology 24:47–171.
iNaturalist contributor (jillcrosthwaite), iNaturalist. 2022. iNaturalist Research-grade Observations. iNaturalist.org. Website: [Accessed April 2022].
Jennings, T.J., and J.P. Barkham. 1979. Litter decomposition by slugs in mixed deciduous woodland. Holarctic Ecology 2:21–29.
Jordan, S.F. and S.H. Black. 2012. Effects of forest land management on terrestrial mollusks: a literature review. USDA Forest Service, Region 6 USDI Oregon/Washington, Bureau of Land Management. 87 pp.
Kimura, K. and S. Chiba. 2010. Interspecific interference competition alters habitat use patterns in two species of land snails. Evolutionary Ecology 24:815–825.
Knapp, E.E., B.L. Estes, and C.N. Skinner. 2009. Ecological effects of prescribed fire season: a literature review and synthesis for managers. USA General Technical Report. Albany, California. 80 pp.
Koprivnikar, J. and P.A. Walker. 2011. Effects of the herbicide Atrazine’s metabolites on host snail mortality and production of trematode cercariae. Journal of Parasitology 97:822–827.
Laskowski, R. and S.P. Hopkin. 1996. Effect of Zn, Cu, Pb, and Cd on fitness in snails (Helix aspersa). Ecotoxicology and Environmental Safety 34:59–69.
Lebourcq, G. 2020. Biologie de la conservation : Comment évaluer et intégrer les exigences écologiques des gastéropodes terrestres cryptiques ? - Le cas de l’espèce protégée Elona quimperiana. Master 1 thesis, University Rennes 1, France, 24 pp.
Lee, H., W. Bakowsky, J. Riley, J. Bowles, M. Puddister, P. Uhlig, and S. McMurray. 1998. Ecological Land Classification for Southern Ontario. SCSS Field Guide FG-02. North Bay: Ontario Ministry of Natural Resources. 225 pp.
Mahlfeld, K. 2000. Impact of introduced gastropods on molluscan communities, northern North Island. Conservation advisory notes no 277, Department of Conservation, Wellington, New Zealand. 18 pp.
Mason, C.F. 1970a. Food, feeding rates and assimilation in woodland snails. Oecologia 4:358–373.
Mason, C.F. 1970b. Snail populations, beech litter production, and the role of snails in litter decomposition. Oecologia 5:215–239.
Michigan Invasive Species. 2022. Invasive Species: Japanese Chaff Flower. Michigan Government website. [Accessed April 2022].
Morand, S., M.J. Wilson, and D.M. Glen. 2004. Nematodes (Nematoda) parasitic in terrestrial gastropods. Pp. 525–558. In: G. Barker (ed.). Natural Enemies of Terrestrial Mollusks. CABI Publishing, Cambridge, Massachusetts.
NatureServe. 2022. NatureServe Explorer [web application]. NatureServe, Arlington, Virginia. [Accessed: April 05, 2022].
NCC (Nature Conservancy of Canada). 2008. Management Guidelines: Pelee Island Alvars. NCC – Southwestern Ontario Region, London, Ontario. 43 pp.
Nekola, J.C. 2010. Acidophilic terrestrial gastropod communities in North America. Journal of Molluscan Studies 76:144–156.
Nicolai, A. 2010. The impact of diet treatment on reproduction and thermos-physiological processes in the land snail Cornu aspersum and Helix pomatia. Thèse de Doctorate en co-tutelle, Universität Bremen, Allemagne/ Université Rennes 1, France, 205 pp.
Nicolai, A., J. Filser, V. Briand, and M. Charrier. 2010. Seasonally contrasting life history strategies in the land snail Cornu aspersum: physiological and ecological implication. Canadian Journal of Zoology 88:995-1002.
Nicolai, A., J. Filser, R. Lenz, C. Bertrand, and M. Charrier. 2011. Adjustment of metabolite composition in the haemolymph to seasonal variations in the land snail Helix pomatia. Journal of Comparative Physiology B 181:457–466.
North-South Environmental Inc. 2004. Vegetation Communities and Significant Vascular Plant Species of Middle Island, Lake Erie. Report for Point Pelee National Park. 94 pp.
Notten, M.J.M., A.J.P. Oosthoek, J. Rozema, and J. Aerts. 2005. Heavy metal concentrations in a soil-plant-snail food chain along a terrestrial soil pollution gradient. Environmental Pollution 138:178–190.
Örstan, A. 2006. Rearing terrestrial gastropoda. Pp. 287–293. In: C.F. Sturm, T.A. Pearce, and A. Valdés (eds.). The Mollusks: A Guide to Their Study, Collection, and Preservation. American Malacological Society, Pittsburgh, Pennsylvania. 445 pp.
Oughton, J. 1948. A Zoogeographical Study of the Land Snails of Ontario. University of Toronto Press, Toronto. 128 pp.
Parks Canada. 2007. Point Pelee National Park of Canada State of the Park Report 2006. Her Majesty the Queen in Right of Canada. Leamington, Ontario. 44 pp.
Parks Canada. 2008. Point Pelee National Park of Canada. Middle Island Conservation Plan 2008-2012. Parks Canada, Leamington, Ontario. 44 pp.
Pearce, T.A. 1990. Spooling and line technique for tracing field movements of terrestrial snails. Walkerana 4:307–316.
Pearce, T.A. and A. Örstan. 2006. Terrestrial gastropoda. Pp. 261–285. In: C.F. Sturm, T.A. Pearce, and A Valdés (eds.). The Mollusks: A Guide to Their Study, Collection, and Preservation. American Malacological Society, Pittsburgh, Pennsylvania. 445 pp.
Pearce, T.A. and M.E. Paustian. 2013. Are temperate land snails susceptible to climate change through reduced altitudinal ranges? A Pennsylvania example. American Malacological Bulletin 31:213–224.
Pilsbry, H.A. 1940. Land Mollusca of North America (North of Mexico). Volume 1, Part 2. Academy of Natural Sciences of Philadelphia, Monograph 3:i–vi + 575–997.
Qiu, J. and M.G. Turner. 2017. Effects of non-native Asian earthworm invasion on temperate forest and prairie soils in the Midwestern US. Biological Invasions 19:73–88.
Reck, H., and R. van der Reer. 2015. Insects, snail and spiders: the role of invertebrates in road ecology. Pp. 247–257. In: R. van der Reer, D.J. Smith, and C. Grilo (eds.). Handbook of Road Ecology. John Wiley & Sons, Oxford, United Kingdom.
Reynolds, J.W. 2011. The earthworms (Oligochaeta: Lumbricidae) of Pelee Island, Ontario, Canada. Megadrilogica 15(3):23–33.
Reynolds, J.W. 2014. A checklist by counties of earthworms (Oligochaeta: Lumbricidae, Megascolecidae and Sparganophilidae) in Ontario, Canada. Megadrilogica 16:111–135.
Robinson, D.G. 1999. Alien invasions: the effects of the global economy on non-marine gastropod introduction into the United States. Malacologia 41:413–438.
Robinson, D., and J. Slapcinsky. 2005. Recent introductions of alien gastropods into North America. American Malacological Bulletin 20:89–93.
Roy, D.B., D.A. Bohan, A.J. Haughton, M.O. Hill, J.L. Osborne, S.J. Clark, J.N. Perry, P. Rothery, R.J. Scott, D.R. Brooks, G.T. Champion, C. Hawes, M.S. Heard, and L.G. Firbank. 2003. Invertebrates and vegetation of field margins adjacent to crops subject to contrasting herbicide regimes in the Farm Scale Evaluations of genetically modified herbicide-tolerant crops. Philosophical Transactions of the Royal Society London B 358:1879–1898.
Rush, S.A., S. Verkoeyen, T. Dobbie, S. Dobbyn, C.E. Hebert, J. Gagnon, and A.T. Fisk. 2011. Influence of increasing populations of Double-crested Cormorants on soil nutrient characteristics of nesting islands in western Lake Erie. Journal of Great Lakes Research 37(2):305–309.
Sandilands, A. 2005. Birds of Ontario: Habitat Requirements, Limiting Factors, and Status. Volume 1. Nonpasserines: Waterfowl through Cranes. University of British Columbia Press. Vancouver, British Columbia. 365 pp.
Schüpbach, H.U., B. Baur. 2008. Parasitic mites influence fitness components of their host, the land snail Arianta arbustorum. Invertebrate Biology 127(3):350-356.
Stoll, P., K. Gatzsch, H. Rusterholz, and B. Baur. 2012. Response of plant and gastropod species to knotweed invasion. Basic and Applied Ecology 13:232-240.
Thorndyke, R. and T. Dobbie. 2013. Point Pelee National Park of Canada. Report on research and monitoring for year 5 (2012) of the Middle Island Conservation Plan. Parks Canada, Leamington, Ontario. 34 pp.
Towne, G., and C. Owensby. 1984. Long-term effects of annual burning at different dates in ungrazed Kansas tallgrass prairie. Journal of Range Management 37:392–397.
Turgeon, D.D., J.F. Quinn, Jr., A.E. Bogan, E.V. Coan, F.G. Hochberg, W.G. Lyons, P. Mikkelsen, R.J. Neves, C.F.E. Roper, G Rosenberg, B. Roth, A. Scheltema, F.G. Thompson, M. Veccione, and J.D. Williams. 1998. Common and Scientific Names of Aquatic Invertebrates from the United States and Canada: Mollusks, Second Edition. American Fisheries Society Special Publication. 26. Bethesda, Maryland. 526 pp.
Utz, R.M., T.A. Pearce, D.L. Lewisa, and J.C. Manninoa. 2018. Elevated native terrestrial snail abundance and diversity in association with an invasive understory shrub, Berberis thunbergia, in a North American deciduous forest. Acta Oecologica 86:66–71.
Varrin, R., J. Bowman, and P.A. Gray. 2007. The known and potential effects of climate change on biodiversity in Ontario’s terrestrial ecosystems: case studies and recommendations for adaptation. Climate Change Research Report CCRR-09. Ontario Ministry of Natural Resources and Forestry. Queen’s Printer for Ontario, Toronto, Ontario. 47. 1379 pp.
Viard, B., F. Pihan, S. Promeyrat, and S.J.-C. Pihan. 2004. Integrated assessment of heavy metal (Pb, Zn, Cd) highway pollution: bioaccumulation in soil, Graminaceae and land snails. Chemosphere 55:1349–1359.
Whitson, M. 2005. Cepaea nemoralis (Gastropoda, Helicidae): the invited invader. Journal of the Kentucky Academy of Science 66:82–88.
Williams, G.W. 2000. Reintroducing Indian type fire: implications for land managers. Fire Management Today 60(3):40–48.
Wirth, T., P. Oggier, and B. Baur. 1999. Effect of road width on dispersal and population genetic structure in the land snail Helicella itala. Journal of Nature Conservation 8:23–29.
Xu, Y., A.J. Li, K. Li, J. Qin, and H. Li. 2017. Effects of glyphosate-based herbicides on survival, development and growth of invasive snail (Pomacea canaliculata). Aquatic Toxicology 193:136–143.
Personal communications
Dobbie, T. 2022. Verbal and e-mail communication. March 8, 2022. Park Ecologist – Nature Legacy, Point Pelee National Park, Parks Canada.