Wild Hyacinth Recovery Strategy
This document advises the ministry on ways to ensure healthy numbers of the Wild Hyacinth, a threatened or endangered species, return to Ontario.
Recovery strategy prepared under the Endangered Species Act, 2007

About the Ontario recovery strategy series
This series presents the collection of recovery strategies that are prepared or adopted as advice to the Province of Ontario on the recommended approach to recover species at risk. The Province ensures the preparation of recovery strategies to meet its commitments to recover species at risk under the Endangered Species Act (ESA) and the Accord for the Protection of Species at Risk in Canada.
What is recovery?
Recovery of species at risk is the process by which the decline of an endangered, threatened, or extirpated species is arrested or reversed, and threats are removed or reduced to improve the likelihood of a species' persistence in the wild.
What is a recovery strategy?
Under the ESA a recovery strategy provides the best available scientific knowledge on what is required to achieve recovery of a species. A recovery strategy outlines the habitat needs and the threats to the survival and recovery of the species. It also makes recommendations on the objectives for protection and recovery, the approaches to achieve those objectives, and the area that should be considered in the development of a habitat regulation. Sections 11 to 15 of the ESA outline the required content and timelines for developing recovery strategies published in this series.
Recovery strategies are required to be prepared for endangered and threatened species within one or two years respectively of the species being added to the Species at Risk in Ontario list. There is a transition period of five years (until June 30, 2013) to develop recovery strategies for those species listed as endangered or threatened in the schedules of the ESA. Recovery strategies are required to be prepared for extirpated species only if reintroduction is considered feasible.
What’s next?
Nine months after the completion of a recovery strategy a government response statement will be published which summarizes the actions that the Government of Ontario intends to take in response to the strategy.
The implementation of recovery strategies depends on the continued cooperation and actions of government agencies, individuals, communities, land users, and conservationists.
For more information
To learn more about species at risk recovery in Ontario, please visit the Ministry of Natural Resources Species at Risk webpage at: www.ontario.ca/speciesatrisk
Recommended citation
Jalava, J.V. 2013. Recovery Strategy for the Wild Hyacinth (Camassia scilloides) in Ontario. Ontario Recovery Strategy Series. Prepared for the Ontario Ministry of Natural Resources, Peterborough, Ontario. v + 26 pp + appendix.
© Queen’s Printer for Ontario, 2013
ISBN 978-1-4435-9435-6 (PDF)
Content (excluding the cover illustration) may be used without permission, with appropriate credit to the source.
Cette publication hautement spécialisée Recovery strategies prepared under the Endangered Species Act, 2007, n'est disponible qu'en Anglais en vertu du Règlement 411/97 qui en exempte l'application de la Loi sur les services en français. Pour obtenir de l'aide en français, veuillez communiquer avec Pamela Wesley au ministère des Richesses naturelles au 705-755-5217.
Authors
Jarmo V. Jalava, Consulting Ecologist, Carolinian Canada Coalition
Acknowledgments
This recovery strategy was prepared by Jarmo Jalava in consultation with the Carolinian Woodlands Plants Technical Committee and with input from: Allen Woodliffe (Ontario Ministry of Natural Resources, Aylmer District); Michael Oldham and Sam Brinker (Ontario Natural Heritage Information Centre); John Ambrose (Botanical Consultant); Chris Risley, Amelia Argue, Vivian Brownell, Carolyn Seburn, Eric Snyder and Bree Walpole (Ontario Ministry of Natural Resources, Species At Risk Branch); Kate Hayes, Graham Bryan, Angela Darwin and Barbara Slezak (Canadian Wildlife Service, Environment Canada); Mhairi McFarlane (Nature Conservancy of Canada); Judith Jones (ecological consultant); Melody Cairns and Sandy Dobbyn (Ontario Parks); Dan Lebedyk (Essex Region Conservation Authority); and Kim Borg and Valerie Minelga (Parks Canada Agency).
Declaration
The recovery strategy for the Wild Hyacinth was developed in accordance with the requirements of the Endangered Species Act, 2007 (ESA). This recovery strategy has been prepared as advice to the Government of Ontario, other responsible jurisdictions and the many different constituencies that may be involved in recovering the species.
The recovery strategy does not necessarily represent the views of all of the individuals who provided advice or contributed to its preparation, or the official positions of the organizations with which the individuals are associated.
The goals, objectives and recovery approaches identified in the strategy are based on the best available knowledge and are subject to revision as new information becomes available. Implementation of this strategy is subject to appropriations, priorities and budgetary constraints of the participating jurisdictions and organizations.
Success in the recovery of this species depends on the commitment and cooperation of many different constituencies that will be involved in implementing the directions set out in this strategy.
Responsible jurisdictions
Ontario Ministry of Natural Resources
Environment Canada – Canadian Wildlife Service, Ontario
Parks Canada Agency
Executive summary
Wild Hyacinth (Camassia scilloides) is a showy, perennial, spring-flowering plant of the lily family (Liliaceae) that develops from a bulb. It has linear leaves and a stalk that supports up to 100 pale blue to white star-shaped flowers. Within its Canadian range, it grows in partial to moderate shade in low, moist woods on clay soil, as well as in drier, scrubbier hawthorn thickets and hackberry woodlands on shallow, rocky soil over limestone bedrock. In the United States the species also occurs in deeper-soiled floodplain forests, prairies, and a variety of other semi-open habitats.
Wild Hyacinth is designated Threatened under the Endangered Species Act (ESA). It was designated as threatened in Canada by COSEWIC in 2002, and this status was adopted federally under the Species at Risk Act. It is of conservation concern or very rare (S1 or S2) in 8 of the 25 states of the United States within its range. However, it is common in its core distribution area, the Mississippi basin.
All extant and historic Ontario occurrences are from islands in western Lake Erie and the Detroit River in southwestern Essex County. Based on targeted surveys in 2001 and subsequent site visits, five extant occurrences are large (2,000 to >5,000 plants) and are believed to be stable. Habitat damage from colonial-nesting Double-crested Cormorants is believed to have destroyed two occurrences within the past two decades, and another was seriously impacted but has recovered since cormorant management was initiated at the site. One sub-population on Pelee Island has also disappeared within the past 15 years due to land clearing. The two extirpated historic populations were also lost to land development.
The recovery goal is to maintain and, where necessary, establish self-sustaining and viable populations of Wild Hyacinth at extant sites and at suitable historical sites in southern Ontario. Although extant occurrences appear to have stable populations, intervention may be necessary to prevent population declines due to cormorant impacts or possible future private land development.
The recovery objectives are to:
- protect and manage habitat to maintain extant populations in Ontario;
- determine current distribution and abundance of Wild Hyacinth populations in Ontario through inventory and monitoring in association with other Carolinian woodland plant species at risk;
- address knowledge gaps relating to their biology, ecology, habitat and threats;
- determine feasibility of reintroduction, and reintroduce Wild Hyacinth populations to suitable historical sites and recovery habitat; and
- prepare and disseminate information on best management practices for Wild Hyacinth.
It is recommended that an occupancy-based approach be used to define the area to be regulated as habitat under the ESA. Given that Wild Hyacinth does not occupy all apparently suitable habitat at the few extant Ontario sites, it is recommended that the area occupied by the plants, as well as surrounding habitat required for dispersal and population expansion, be prescribed as habitat in the regulation. The area prescribed should be delineated as the full extent of the Ecological Land Classification (ELC) ecosite polygon (as mapped by a qualified biologist, ecologist, or equivalent) within which a population occurs.
Wild Hyacinth is occasionally cultivated. It is recommended that horticultural populations be excluded from the habitat regulation.
1.0 Background information
1.1 Species assessment and classification
Common name: Wild Hyacinth
Scientific name: Camassia scilloides
SARO List Classification: Threatened
SARO List History: Threatened (2004)
COSEWIC Assessment History: Threatened (2002), Special Concern (1990)
SARA Schedule 1: Threatened
Conservation status rankings:
GRANK: G4G5
NRANK: N2
SRANK: S2
The glossary provides definitions for technical terms, including the abbreviations above.
1.2 Species description and biology
Species description
Wild Hyacinth (Camassia scilloides) is a showy, perennial, spring-flowering plant of the lily family (Liliaceae) (Gould 1942). It develops from a bulb and has basal, keeled, linear leaves. It has a stalk up to 70 cm tall that supports up to 100 star-shaped flowers, pale blue to white in colour, with six petals and yellow anthers (Gould 1942, Gleason and Cronquist 1991).
Species biology
In Canada, Wild Hyacinth blooms in mid to late May when pollinating insects are abundant. Insect visitors that have been observed on sunny days include butterflies (superfamily Papilionoidea), bumblebees (Bombus spp.), metallic green/bronze bees (Agapostemon spp.), small solitary bees (Megachilidae), bee flies (Bombiliidae) and hover flies (Syrphidae), but little or no insect visitation occurred during cloudy or rainy weather (COSEWIC 2002). As with many other spring ephemerals, leaves and fruiting stalks die down by mid-summer.
Germination occurs the following spring. Seeds are produced copiously from dry capsules, some of which fall over before opening, suggesting that seeds are not dispersed very far. The dry, hard seeds do not seem to be attractive to woodland dispersal agents, such as ants. Within a habitat patch there may be several densely- clustered Wild Hyacinth colonies. It is uncertain what dispersal mechanisms are at play in creating a new colony (COSEWIC 2002).
It appears that vegetative reproduction from bulb offsets does not occur frequently in Canadian populations (Oldham 1990, COSEWIC 2002). During field work for COSEWIC (2002), a group of Wild Hyacinths was dug up to determine if there were indications of vegetative growth of populations. No evidence of offshoots or bulblets was seen. However, numerous small bulbs were among (but clearly separate from) the mature bulbs, suggesting seedlings of different ages. The occurrence of mixed flower colours within colonies (white to very pale blue to pale blue) is further indication of genetic variation that would not be expected in populations that have developed by vegetative means (COSEWIC 2002).
Individual plants are likely long-lived, since the closely related Common Camas (Camassia quamash) of western North America has a lifespan of 15 to 20 years (Stevens et al. 2001). There has been little change in location from different observations of Wild Hyacinth in Ontario over the decades (COSEWIC 2002).
Wild Hyacinth has an observed interaction with a variety of pollinating insects, a relationship that undoubtedly is symbiotic, providing food energy to the pollinators and an exchange of genetic material for the plants. As a staple native food plant of North American Aboriginal people, hyacinths (Camassia spp.) played a significant socio-ecological role in functioning ecosystems prior to European settlement (e.g., see Garibaldi and Turner 2004).
1.3 Distribution, abundance and population trends
Wild Hyacinth occurs naturally in the southeastern and midwestern United States, with its distribution centred on the Mississippi drainage basin. It is found from northwestern Georgia to eastern Texas and north to its extremely limited range in Canada, on the Lake Erie Islands of Ontario. Western Lake Erie and southern Wisconsin mark the northeastern and northwestern limits of its range, respectively.
Wild Hyacinth is of conservation concern in nearly all the state and provincial jurisdictions within which its status has been assessed. It is designated endangered in Pennsylvania and Wisconsin and threatened in Michigan and North Carolina (USDA 2012). It is also classified as threatened in Canada under the Species at Risk Act and in Ontario under the Endangered Species Act, 2007. According to NatureServe (2012), it is ranked "critically imperilled" (S1) in North Carolina and Pennsylvania, "imperilled" (S2) in Ontario, Georgia, Iowa, Michigan, South Carolina, Virginia and Wisconsin, "imperilled to vulnerable" (S2S3) in Mississippi, "vulnerable" (S3) in Louisiana and "vulnerable to apparently secure" (S3S4) in Illinois. It is considered "apparently secure" (S4) in two states, Kentucky and West Virginia (NatureServe 2012). Its conservation status has not been ranked in Missouri, Kansas, Tennessee, Texas and Alabama, according to NatureServe (2012)
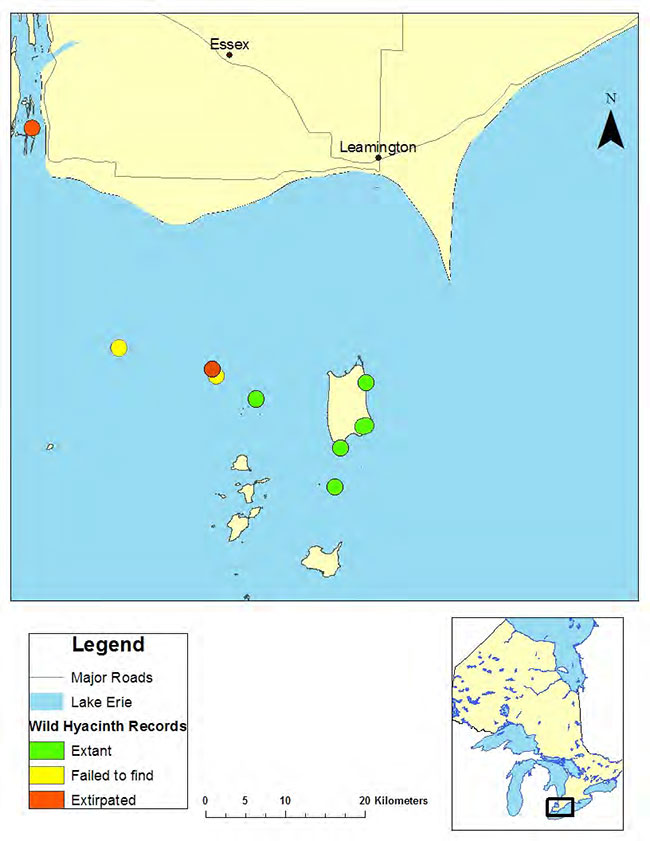
All extant and historic Ontario occurrences of Wild Hyacinth are from islands in Lake Erie and the Detroit River in Essex County (COSEWIC 2002, NHIC 2006). One historic record of Wild Hyacinth on the mainland of Point Pelee National Park is believed to have been planted (PCA 2011a). Based on detailed surveys for the species between 2001 (COSEWIC 2002) and 2012 (PCA 2011b, McFarlane (pers. comm. 2012), five extant occurrences are relatively large (2,000 to >5,000 plants). Due to impacts from colonial-nesting Double-crested Cormorants (Phalacrocorax auritus), the East Sister Island and Middle Sister Island populations appear to no longer be extant. The Middle Island population was reduced from ~5,000 individuals in 1988 to less than 865 plants (700 of them vegetative) in 2001, and no more than 500 in 2003 (North-South Environmental 2004), before recovering to approximately 4,000 plants in 2008, the year cormorant population management was begun at the site.
There is no evidence that Wild Hyacinth was ever common in Ontario. In the late 1980s, the total estimated population for the province was between 14,000 to 16,000 plants (Oldham 1990), not including a large population on Hen Island for which no count was available. A 2001 survey of all known sites, undertaken with a crew of knowledgeable volunteers, yielded a total count of 21,212 flowering plants (COSEWIC 2002), suggesting relatively stable populations in recent decades at most extant sites. However, the loss of the East Sister Island population apparently to cormorant impacts (COSEWIC 2002), combined with the loss of two historical populations and a Fish Point sub-population to land development, suggests a long term overall decline of the species in the province.
Hyacinth (Camassia spp.) bulbs are starchy and edible, and the western North American species (C. leichtlinii and C. quamash) was consumed as a staple food by native people and by early European explorers. Wild Hyacinth bulbs were probably similarly used by eastern Aboriginal people in the Lake Erie region. It is possible that the species was deliberately or accidentally introduced to the Erie Islands by them (COSEWIC 2002), which may offer clues as to the species' ability to persist and/or expand its populations in Ontario. Hyacinth bulbs, usually of the western species, are commonly available in the specialty horticultural trade (COSEWIC 2002), which could affect the "purity" of the native populations should exchange of genetic material occur.
Table 1. Extant and Historic Occurrences of Wild Hyacinth in Ontario (based on COSEWIC 2002, PCA 2011b, NHIC 2012, McFarlane pers. comm. 2012).
| Ontario Population County or Region |
Ontario Population Site |
Quality rank* | Last Obs. | Status | Notes |
|---|---|---|---|---|---|
| Essex | Hen Island, Lake Erie | A** | 2001 | One population; 5680 plants | Private land; invasive species a threat. |
| Essex | Middle Point | A | 2011 | Two sub-populations; 4862 plants in 2001; population considered similar or slightly larger in 2011. | Owned by Nature Conservancy of Canada (NCC); population appears thriving, no immediate threats noted. Previously undocumented population of 10-15 plants on private land east of East Shore Road may be threatened by Periwinkle (Vinca minor) (McFarlane pers. comm. 2012). |
| Essex | Stone Road Alvar | A | 2001 | Five or six sub-populations (Woodliffe pers. comm. 2011); 4485 plants | Most of site protected as nature reserve by various agencies; populations on private land possibly threatened by development. |
| Essex | Fish Point Provincial
Nature Reserve |
A | 2001 | Two sub-populations; 2090 plants | One private-land sub-population extirpated by housing development; others protected within Fish Point Provincial Nature Reserve. |
| Essex | Middle Island | B | 2008 | ~4000 plants (PCA 2011b) | Acquired by NCC in 2001, now managed by Point Pelee National Park; Double-crested Cormorant being managed to reduce impacts. |
| Essex | Middle Sister Island | F*** | 2001, 2011 | One sub-population; 3230 plants in 2001. None observed in 2011. | Private land; possibly threatened by development. Incidental observations in 2011 indicate that population is likely gone; island covered in coromorant guano with only remaining herbaceous vegetation being a stand of Pokeweed (Phytolacca americana) (PCA 2011b). |
| Essex | East Sister Island | H**** | 1985 | No plants found in 2001, presumed extirpated | Provincial Nature Reserve; habitats heavily impacted by Double-crested Cormorant nesting colony. |
| Essex | North Harbour Island | X | 1948 | Presumed extirpated | Private land. Most of the island is now an extensive lawn; detailed searches in 1987 failed to locate the species. |
| Essex | Bois-blanc Island, Detroit River ("White Island, west of Amherstberg) | X | 1882 | Presumed extirpated | Private land. All recent (to 1999 at least) searches have been unsuccessful; most of the island is now an amusement park (COSEWIC 2002). |
* Quality Rank refers to Natural Heritage Information Centre (NHIC) ranks based on predicted viability of the occurrence: A – Excellent, B – Good, C – Fair, D – Probably not viable; E – Verified extant, F – Failed to find, H – Historic, X – Extirpated
** status assigned as "E" by NHIC (2006) requires update to "A"
*** status assigned as "A" by NHIC (2006) requires update to "F"
**** status assigned as "C" by NHIC (2006) requires update to "H".
Figure 1. Historical and current distribution of Wild Hyacinth in Ontario
1.4 Habitat needs
Wild Hyacinth is restricted in Canada to islands at the west end of Lake Erie, an area with a climate moderated by this large body of water. This region has one of the longest growing seasons in Ontario and the highest heat units in Canada, as well as relatively dry summers compared to other parts of southern Ontario. Within this restricted range, Wild Hyacinth grows in the partial to moderate shade of open deciduous woodlands and hawthorn scrub. The rich clayey Farmington loam (Richards et al. 1949) ranging to organic soils in these habitats is shallow, with limestone bedrock occurring close to the surface (COSEWIC 2002).
In Ontario, the extant occurrences of Wild Hyacinth are in low, moist woods on clay soil as well as drier scrubbier woodland on shallow, rocky soil over limestone bedrock (Oldham 1990). The Stone Road Alvar occurrences are found in shrubby alvar, Blue Ash – Chiquapin Oak – Hackberry savannah, Red Ash – Swamp Oak forest, Shagbark Hickory forest, and Hackberry – Hawthorn savannah. At Middle Point, habitats in which the species occurs are described as Hackberry – Maple forest, Hackberry forest, rich Hackberry forest, moist woods and rocky woods. Tree and shrub dominants at other occurrences include Red Ash (Fraxinus pennsylvanica), Downy Hawthorn (Crataegus mollis) and Common Hackberry (Celtis occidentalis), with overstorey associates such as Shagbark Hickory (Carya ovata), Bur Oak (Quercus macrocarpa) and Sugar Maple (Acer saccharum) (Oldham 1990). Herbaceous associates at these sites include False Mermaid (Floerkia proserpinacoides), Canada Avens (Geum canadense), Spring Avens (Geum vernum), Garlic Mustard (Alliaria petiolata), Kidney-leaved Buttercup (Ranunculus abortivus), Virginia Creeper (Parthenocissus sp.), Spotted Jewelweed (Impatiens capensis), Long-styled Sweet-cicely (Osmorhiza longistylis), Cleavers (Galium aparine), Blue Phlox (Phlox divaricatus), Short’s Aster (Aster shortii), Canada Onion (Allium canadense), White Trout-lily (Erythronium albidum) and Virginia Waterleaf (Hydrophyllum virginianum) (Oldham 1990). The habitat of the historic Bois Blanc Island occurrence on the Detroit River was described as "wet meadows".
In the United States, the species also occurs in deeper-soiled floodplain forests, and in Wisconsin it is considered a species of "damp prairie soils, roadsides and railroad right-of-ways" (WDNR 2006), while in Missouri it is a plant of "prairies, glades and open woods" (CCM 2006). Oldham (1990) lists the following habitat types for United States occurrences, based on various literature sources and herbarium data: prairie meadow; slightly dry to wet prairies; open woods; calcareous glades or outcrops in rocky forests; moist woods or meadows; hill prairies and railroad prairie; edges of woods, particularly in calcareous areas; floodplain woods and along streams; steep, rocky calcareous wooded slopes; hackberry woods; river-bottom flats and banks; roadside wet-mesic prairie remnant; wet meadows; grassy road bank; oak woods on steep slope; limestone hillside; low oak – red cedar – hackberry woods; lowest and wettest spots in prairie; low granite ridges, prairie; rich, moist woods.
In horticultural settings, Camassia are also tolerant of a wide range of habitats and moisture levels, and do well in any fertile soil that is moist but not saturated in spring (Oldham 1990). Plants of this genus do not appear to be negatively impacted by droughty conditions in summer, and appear to do best in full sun (Oldham 1990), although preference for "part shade" or "shade" is cited as the light requirement in UTLBJWC (2012).
1.5 Limiting factors
Wild Hyacinth is at the extreme northern limit of its range on the Lake Erie Islands, where temperature extremes are reduced and growing season extended because of the moderating effect of the adjacent waters. This suggests the possibility that climate may be a factor limiting its expansion in Ontario. Since a native population has never been confirmed on the mainland
Macior (1978) suggests that Wild Hyacinth is completely dependent on insects for pollination. This would be a serious limiting factor should its pollinator populations be in decline.
Although it has a widespread geographic distribution, the fact that Wild Hyacinth is considered rare in many jurisdictions (NatureServe 2012) within its range suggests that it may have certain as yet unknown biological or habitat limitations. At the same time, the fact that it can thrive along roadsides and railway lines in some portions of its range (WDNR 2006) suggests that Wild Hyacinth can benefit from moderate levels of certain types of disturbance if other ecological factors are favourable.
1.6 Threats to survival and recovery
Known threats
Hyperabundant Native Species
One of the main threats to the survival of Wild Hyacinth in Canada involves the explosion of Double-crested Cormorant populations on the Lake Erie since the early 1980s. Canada Goose populations have also increased dramatically in southern Ontario during this period (CWS 2012). Cormorants nest in large colonies (more than 5,000 individuals), severely impacting both the trees and the ground flora in their vicinity. Trampling by Canada Geese has been noted as a significant recent impact at the Middle Island population (PCA 2011b). Browsing by geese has also been observed (Woodliffe pers. comm. 2009), although the severity of impact is unknown. Compounding the goose-impacts, ammonia-rich excrement produced by cormorants has destroyed much of the native understorey, creating conditions suitable for rapid colonization by highly-competitive plant species such as Pokeweed (Phytolacca americana) and European Stinging-nettle (Urtica dioica ssp. dioica) (Jalava et al. 2008b). However, recent efforts to control cormorants and deter Canada Geese at Middle Island appear to have resulted in a significant recovery of Wild Hyacinth at the site (PCA 2011b). Cormorants prefer to nest on small, isolated islands with fewer predators, so presumably the Hen Island population would be much more likely to be impacted than the occurrences on Pelee Island.
The increase in Double-crested Cormorants is a recent phenomenon with underlying anthropogenic causal factors. Their expansion into the Great Lakes region from the west in the early 1900's was probably related to an increase in food supply (prey fish species) associated with declines in predatory fish species caused by commercial over- fishing, as well as the introduction of Sea Lamprey, and other factors (Environment Canada 2006). A dramatic population decline of cormorants occurred between the 1950s and 1970s due to bio-accumulating toxic chemicals combined with direct persecution by fishermen who believed cormorants were predating juveniles of desirable commercial and sport fish species. A gradual recovery of populations began in the 1970s after regulations were put in place to control the production and use of DDT and related chemicals in North America, with rapid increases starting in the late 1980s. The United States federal protection under the Migratory Bird Treaty Act in 1972, combined with the expansion of the aquaculture industry and construction of reservoirs in the southeast United States are considered important factors in the resurgence (USFWS 2012). The introduction of Round Goby and other exotic prey species to the lower Great Lakes may also have benefitted cormorants in recent decades. Also, the reduced turbidity of lakes due to the effects introduced mussels has likely made their prey easier to see.
Land development
Two historic Wild Hyacinth occurrences (Bois Blanc and North Harbour islands) were extirpated by clearing for housing and other development (COSEWIC 2002). The habitat of one population at Fish Point (Mosquito Point) on Pelee Island on land zoned as cottage residential was cleared for development prior to Wild Hyacinth’s designation as threatened. Occurrences on private land at Stone Road and Middle Point are not situated near the shoreline where cottage development is more likely, are almost entirely now on private or public conservation lands, and are therefore not likely to be impacted by this threat.
Exotic or invasive species
Invasion of Wild Hyacinth habitat by exotic species is often linked to other threats, such as cormorant colonies, land clearing for development, forestry practices, nearby trails and roads, and other disturbances. Invasive exotic plant species compete for limited resources with native taxa. Their presence alters the overall composition of ecological communities, potentially affecting other taxa, including pollinating insects. Invasive species have been observed as being a problem at both the Middle Island and the (probably extirpated) East Sister Island occurrences (COSEWIC 2002, Jalava et al. 2008b). Garlic Mustard and Norway Maple have been noted as invading woodlands near the Middle Point population (COSEWIC 2002), although not at the occurrence itself, and invasive species control is one of the management priorities at this site (McFarlane pers. comm. 2012). The western Camassia species have been "severely impacted" by introduced species in the Garry Oak ecosystems of Vancouver Island (Garibaldi and Turner 2004).
Emerald Ash Borer has recently arrived on Pelee Island, and the resulting die-back of various ash species has been significant. Ash is one of the most dominant species on Pelee Island, but with the die-back forest canopies are opening up and shrub and vine (especially Parthenocissus spp., Rubus spp., Rhus spp., Smilax spp.) populations have increased greatly. Impacts on the light levels in the understorey may be detrimental to Wild Hyacinth at affected sites (Anonymous 2011).
Potential threats
Trampling
Trampling of plants by humans has not been cited as a threat to Wild Hyacinth in Canada. However, the potential exists at any accessible site on Pelee Island, even though venturing off trails may be prohibited at Fish Point Provincial Nature Reserve and other protected sites. The amount of visitation by boaters to outlying smaller Erie Island sites is not known, but it is probably relatively infrequent. Trampling may cause direct damage to plants and disturb the habitat, creating conditions suitable for invasion by exotic species. At present this should be considered a relatively minor threat. As noted above, trampling by Canada Geese was damaging Middle Island populations until goose deterrents were put in place (Anonymous 2011, PCA 2011b).
Collecting for horticulture
Wild Hyacinth is a showy, attractive, spring-flowering plant; and the potential for its collection by horticultural enthusiasts is considerable. Hyacinth (Camassia spp.) are widely available in the specialty horticultural trade, although the western species and related cultivars are most frequently sold commercially in North America (COSEWIC 2002). Given that there are no documented instances of Wild Hyacinth collecting in Ontario, at present this is also considered a relatively minor threat.
Natural succession
Given that all Ontario populations of Wild Hyacinth occur in open or semi-open woodland habitats, near openings, or along edges, probably due to light requirements, natural succession to closed-canopy conditions could render habitat unsuitable for the species. However, some of the larger Ontario populations occur away from forest edges, and have quite extensive canopy cover by late spring and throughout the summer. A dominant tree species at these sites is Common Hackberry (Celtis occidentalis), one of the last of the tree species to leaf out, thereby allowing sunlight to reach the forest floor throughout the peak flowering period. By the time heavy shade takes over, flowering is complete, and seed set is largely established (Woodliffe pers. comm. 2011).
1.7 Knowledge gaps
Additional study required to better understand the status and ecology of Wild Hyacinth in Ontario includes:
- determining what constitutes a viable, self-sustaining population in Ontario;
- determining through field surveys and monitoring the most current distribution and population sizes of extant populations, particularly at heavily impacted sites;
- better understanding of population trends province-wide and at individual sites;
- determining if current recommended management practices for Double-crested Cormorant colonies are appropriately addressing the recovery needs of Wild Hyacinth populations at impacted sites;
- better definition of the habitat requirements for this species in Ontario;
- determining the degree of threat by adjacent land development and forest management on remaining private land sites;
- better understanding of the relative severity and importance of potential and poorly-understood threats such as invasive species, trampling and collection for horticulture;
- better understanding of Wild Hyacinth reproductive biology (seed dormancy, viability and germination; seed bank structure; pollination methods and pollinators), dispersal (mechanisms and distances) and establishment (ecological conditions, minimum propagule pressure) would be beneficial;
- best management practices are largely undocumented (e.g., see Franken et al 2009, on potential effects of prescribed burns); and
- understanding establishment requirements and feasibility (prior to reintroduction to historic sites).
1.8 Recovery actions completed or underway
A comprehensive survey of known extant and historic Wild Hyacinth populations was undertaken in 2001 by a team of volunteers (COSEWIC 2002).
Passive management is the standard practice at Provincial Nature Reserves (East Sister Island and Fish Point), although specific management for Wild Hyacinth is not noted in the management plan for Fish Point (Ontario Parks 2005), in which the species is erroneously indicated as being designated Special Concern. Stone Road Alvar is a nature reserve with portions owned by the Federation of Ontario Naturalists, the Nature Conservancy of Canada (NCC) and the Essex Region Conservation Authority (ERCA); on both the Ontario Nature and ERCA properties, management efforts have included prescribed burning to prevent the natural succession of shrubs from closing in on the savannah communities (Ontario Nature 2006).
Middle Island is managed for natural heritage protection by Point Pelee National Park. A control program for the Double-crested Cormorant was initiated in 2008 with the aim of reducing impacts of the nesting colony on the site’s vegetation and associate species at risk.
The NCC's Natural Area Conservation Plan for the Western Erie Islands includes Wild Hyacinth habitat as one of its top priority conservation targets. NCC recently acquired the property with the major population at Middle Point, as well as one of the sub- populations at Stone Road Alvar, and is undertaking surveys, monitoring and management for Wild Hyacinth at these sites (McFarlane pers. comm. 2012).
In 2007, Waldron (pers. comm. 2012) conducted an inventory of the Bois-blanc Island’s flora and fauna. No Wild Hyacinth was observed during that season’s fieldwork. However, areas in the south portion of the island would likely support a population of Wild Hyacinth. Some of these areas are under Federal control although Essex Region Conservation Authority is responsible for their management. According to Waldron (pers. comm. 2012) this area may be a suitable repatriation site.
2.0 Recovery
2.1 Recovery goal
The recovery goal is to maintain and, where necessary, establish self-sustaining and viable populations of Wild Hyacinth at extant sites and at suitable historical sites in southern Ontario.
At a minimum, several robust, reproducing populations are needed to ensure long-term viability in Ontario. Maintenance of extant populations at their current levels, with no further habitat loss, along with re-establishment of the East Sister Island and Middle Sister Island populations, if restoration of recovery habitat
2.2 Protection and recovery objectives
Table 2. Protection and recovery objectives
| No. | Protection or Recovery Objective |
|---|---|
| 1 | Protect and manage habitat to maintain extant populations in Ontario. |
| 2 | Determine current distribution and abundance of Wild Hyacinth populations in Ontario through inventory and monitoring in association with other Carolinian woodland plant species at risk. |
| 3 | Address knowledge gaps relating to the biology, ecology, habitat and threats. |
| 4 | Determine feasibility of reintroduction, and reintroduce Wild Hyacinth populations to suitable historical sites and recovery habitat. |
| 5 | Prepare and disseminate information on best management practices for Wild Hyacinth. |
2.3 Approaches to recovery
Table 3. Approaches to recovery of the Wild Hyacinth in Ontario
1. Protect and manage habitat to maintain extant populations in Ontario
| Relative Priority | Relative Timeframe | Recovery Theme | Approach to Recovery | Threats or Knowledge Gaps Addressed |
|---|---|---|---|---|
| Critical | Short term | Management | 1.1 Apply, and adapt as necessary, appropriate management practices to reduce cormorant impacts. |
|
| Necessary | Short term | Protection | 1.2 Identify the positive and/or negative impacts of land-use and management practices. |
|
| Necessary | Short term | Protection | 1.3 Identify sites that may be affected by future land development, and ensure that habitat is appropriately mapped and protected through regulation, stewardship and/or securement. |
|
| Necessary | Short term | Protection | 1.4 Develop Best Management Practices (BMPs) for maintaining Wild Hyacinth habitat. |
|
| Necessary | Short term | Protection | 1.5 Encourage and support private land stewards and public land managers to implement BMPs. |
|
| Beneficial | Short term | Management | 1.6 Clarify land ownership of some populations in order to determine stewardship and/or securement options. |
|
| Beneficial | Long term | Stewardship | 1.7 Determine feasibility of restoration or rehabilitation of habitat at historic occurrences and recovery habitat. |
|
| Beneficial | Long term | Stewardship | 1.8 Determine and apply best methods for exotic and invasive species control at impacted sites. |
|
| Beneficial | Long term | Stewardship | 1.9 Integrate restoration planning and recovery actions with NCC's Natural Area Conservation Plan initiative and other programs of partner agencies and groups. |
|
| Beneficial | Long term | Protection | 1.10 Identify key sites for securement in the context of the overall Carolinian Woodlands Recovery Strategy (Jalava et al. 2008a, 2009). |
|
| Beneficial | Long term | Protection | 1.11 Secure key sites through easements or purchase. |
|
2. Determine current distribution and abundance of Wild Hyacinth populations in Ontario through inventory and monitoring in association with other Carolinian woodland plant species at risk
| Relative Priority | Relative Timeframe | Recovery Theme | Approach to Recovery | Threats or Knowledge Gaps Addressed |
|---|---|---|---|---|
| Critical | Short term | Inventory, Monitoring and Assessment | 2.1 Conduct population counts of extant populations every 3 to 5 years, characterize habitat, and assess threats. |
|
| Necessary | Short term | Inventory, Monitoring and Assessment | 2.2 Identify and survey any additional sites with potentially suitable habitat. |
|
| Necessary | Short term | Inventory, Monitoring and Assessment | 2.3 Develop monitoring strategy for Wild Hyacinth (Appendix 1). |
|
| Necessary | Ongoing | Inventory, Monitoring and Assessment | 2.4 Apply the monitoring protocol every 3 to 5 years in association with monitoring for other priority species of the overall Carolinian Woodlands Recovery Strategy (Jalava et al. 2008a, 2009). |
|
3. Address knowledge gaps relating to the biology, ecology, habitat and threats
| Relative Priority | Relative Timeframe | Recovery Theme | Approach to Recovery | Threats or Knowledge Gaps Addressed |
|---|---|---|---|---|
| Critical | Short term | Research | 3.1 Determine the degree to which Double-crested Cormorant colonies must be managed in order to ensure Wild Hyacinth viability. |
|
| Necessary | Short term | Research | 3.2 Assess whether natural succession is impacting populations. |
|
| Necessary | Long term | Research | 3.3 Engage the academic community to investigate Wild Hyacinth reproductive biology (seed dormancy, viability and germination; seed bank structure; pollination methods and pollinators), dispersal (mechanisms and distances) and establishment (ecological conditions, minimum propagule pressure)4. |
|
| Necessary | Short term | Inventory, Monitoring and Assessment | 3.4 Conduct population viability analysis. |
|
| Necessary | Short term | Research, Stewardship | 3.5 Research and identify best management practices for Wild Hyacinth and its habitat. |
|
| Beneficial | Long term | Research | 3.5 Determine if Wild Hyacinth is at risk from collecting by horticulturalists. |
|
| Beneficial | Long term | Education and Outreach | 3.6 Develop educational and outreach materials if necessary, and provide to horticultural clubs and native plant nurseries. |
|
4. Determine feasibility of reintroduction, and reintroduce Wild Hyacinth populations to suitable historical sites and recovery habitat
| Relative Priority | Relative Timeframe | Recovery Theme | Approach to Recovery | Threats or Knowledge Gaps Addressed |
|---|---|---|---|---|
| Beneficial or necessary | Long term | Stewardship | 4.1 Based on assessments of threats, studies of the species' biology and ecology, population viability analysis, determine the need and feasibility of reintroduction. |
|
| Beneficial or necessary | Long term | Stewardship | 4.2. Reintroduce Wild Hyacinth to historic sites where feasible. |
|
5. Prepare and disseminate information on best management practices for Wild Hyacinth
| Relative Priority | Relative Timeframe | Recovery Theme | Approach to Recovery | Threats or Knowledge Gaps Addressed |
|---|---|---|---|---|
| Necessary | Long term | Stewardship | 5.1 Prepare "best management practices" (BMP) fact sheets and provide to landowners and land managers with Wild Hyacinth habitat, as well as to horticultural clubs and garden centres. |
|
4 Ambrose (2012), former curator of the University of Guelph Arboretum, has grown abundant seedlings in a woodland nursery from fall planted seeds and suggests that establishment, at least under controlled conditions, can be undertaken with no apparent difficulty. As a plant of horticultural and reintroduction interest, establishment information is quite widely available (e.g., Horvath et al. 2001, UTLBJWC 2012)
Narrative to support approaches to recovery
Up-to-date information on population size, fruiting and seedling counts, longevity, descriptions of habitat, and assessments of habitat condition and threats, is required at all extant sites in order to prioritize recovery activities. The current status of all populations should be systematically verified.
To maximize efficiency and reduce costs, inventory and recovery actions should be planned and, where appropriate, undertaken in concert with those for other priority species of the overall Carolinian Woodlands Recovery Strategy (Jalava 2008a, 2009). For example, inventory and monitoring for several species occurring at nearby sites would most effectively be undertaken by the same surveyor(s) as part of the same project. Approaches to site management, stewardship and threat mitigation should be developed in association with other Carolinian woodland species at risk that have similar habitat requirements or face similar threats. Similarly, identification of priority sites for restoration or securement should be made using a gap analysis approach that considers the full suite of priority species of the Carolinian Woodlands Recovery Strategy (Jalava 2008a, 2009).
2.4 Performance measures
Evaluation of the overall recovery effort will be measured by the following criteria.
- No loss of extant populations. Populations are increasing or stable (i.e., within an acceptable range of annual fluctuation).
- Habitat is identified and mapped.
- Communications products produced and distributed to landowners and land managers.
- Historic reports and other potential habitat comprehensively surveyed.
- Potential restoration sites identified.
- No further population declines due to cormorant impacts or anthropogenic disturbance (as determined from monitoring data), and threats are being appropriately addressed.
- Habitat restoration initiated where feasible.
- Reintroduction, if feasible, initiated at suitable or restored historic sites.
Evaluation of specific actions taken to recover Wild Hyacinth populations and their Carolinian woodlands habitat will be measured against specific steps and anticipated effects. Evaluation will involve determining whether the action was actually undertaken as prescribed and whether the anticipated effect of the action was realized.
2.5 Area for consideration in developing a habitat regulation
Under the ESA, a recovery strategy must include a recommendation to the Minister of Natural Resources on the area that should be considered in developing a habitat regulation. A habitat regulation is a legal instrument that prescribes an area that will be protected as the habitat of the species. The recommendation provided below by the author will be one of many sources considered by the Minister when developing the habitat regulation for this species.
Wild Hyacinth has a very limited distribution in Ontario, with only six or fewer known extant occurrences, all of them on the islands of western Lake Erie. Because of the extremely low number of occurrences, it is recommended that a precautionary approach be applied in defining habitat for Wild Hyacinth.
Given that the species uses a wide variety of habitats both in Ontario and throughout its range, it is recommended that an occupancy-based approach rather than a generic habitat definition be used to define the area to be regulated. Because Wild Hyacinth does not occupy all apparently suitable habitat and it is extant at fewer than six sites, it is recommended that the regulated area include enough suitable habitat to allow for dispersal and population expansion. It should also be large enough to ensure that direct impacts and adjacent human activities do not negatively affect populations.
The area occupied by Wild Hyacinth plants, as well as surrounding habitat required for dispersal and population expansion, should be prescribed as habitat in the regulation. The area prescribed should be delineated by full extent of the Ecological Land Classification (ELC) ecosite (Lee et al. 1998, Lee 2012) polygon (as mapped by a qualified biologist, ecologist, or equivalent) within which a population occurs.
Defining habitat at the ecosite rather than the more refined ecotype level is precautionary in that it recognizes the need to better understand the habitat requirements of the species. As new information on habitat requirements and site- specific characteristics become available, these attributes should be used to refine the habitat definition, perhaps to the ecotype level. Population viability analysis and new information on reproductive biology and dispersal requirements may also improve the understanding and definition of habitat requirements. In particular, if it is demonstrated that a different areal extent is necessary to allow for dispersal and population expansion, the habitat regulation should be revised to reflect this.
Historic sites have been extirpated primarily due to habitat loss, but there nevertheless appears to be suitable unoccupied habitat within the range of Wild Hyacinth in Ontario. It is therefore recommended that the area described as habitat under the ESA for Wild Hyacinth be flexible enough to allow for repatriation sites, where feasible, should repatriation be deemed necessary or beneficial to recovery.
Wild Hyacinth is occasionally cultivated. It is recommended that horticultural populations be excluded from the habitat regulation.
Glossary
Anthropogenic: Caused or produced by humans.
Committee on the Status of Endangered Wildlife in Canada (COSEWIC): The committee responsible for assessing and classifying species at risk in Canada.
Committee on the Status of Species at Risk in Ontario (COSSARO): The committee established under section 3 of the Endangered Species Act, 2007 that is responsible for assessing and classifying species at risk in Ontario.
Conservation status rank: A rank assigned to a species or ecological community that primarily conveys the degree of rarity of the species or community at the global (G), national (N) or subnational (S) level. These ranks, termed G-rank, N-rank and S-rank, are not legal designations. The conservation status of a species or ecosystem is designated by a number from 1 to 5, preceded by the letter G, N or S reflecting the appropriate geographic scale of the assessment. The numbers mean the following:
1 = critically imperilled
2 = imperilled
3 = vulnerable
4 = apparently secure
5 = secure
Endangered Species Act, 2007 (ESA): The provincial legislation that provides protection to species at risk in Ontario.
Occurrence: An area of land and/or water where a species is, or was, present and has practical conservation value.
Propagule: Any plant material used for the purpose of plant propagation. In asexual reproduction, a propagule may be a woody, semi-hardwood, or softwood cutting, leaf section, or any number of other plant parts. In sexual reproduction, a propagule is a seed or spore.
Species at Risk Act (SARA): The federal legislation that provides protection to species at risk in Canada. This act establishes Schedule 1 as the legal list of wildlife species at risk to which the SARA provisions apply. Schedules 2 and 3 contain lists of species that at the time the Act came into force needed to be reassessed. After species on Schedule 2 and 3 are reassessed and found to be at risk, they undergo the SARA listing process to be included in Schedule 1.
Species at Risk in Ontario (SARO) List: The regulation made under section 7 of the Endangered Species Act, 2007 that provides the official status classification of species at risk in Ontario. This list was first published in 2004 as a policy and became a regulation in 2008.
Symbiotic: A close, prolonged association between two or more different organisms of different species that may, but does not necessarily, benefit each member.
References
Ambrose, J. 2012. Personal communications with J. Jalava and review of draft. Botanical Consultant, Pelee Island, Ontario.
Anonymous. 2011. Jurisdictional review of an earlier draft of this recovery strategy by Ontario Ministry of Natural Resources, Canadian Wildlife Service and Parks Canada Agency staff.
Bickerton, H. 2003. (Draft) Monitoring Protocol for Pitcher’s Thistle (Cirsium pitcheri) – Dune Grasslands. Pitcher’s Thistle – Lake Huron Dune Grasslands Recovery Team. Manuscript.
BONAP. 2012. BONAP's North American Plant Atlas. http://www.bonap.org/BONAPmaps2010/Camassia.html (accessed February, 2012).
Canadian Wildlife Service. 2012. Canada Geese in Southern Ontario. On-line brochure: http://publications.gc.ca/collections/Collection/CW66-251-2005E.pdf (accessed February 2012).
CCM (Conservation Commission of Missouri). 2006. http://www.conservation.mo.gov/nathis/plantpage/flora/wildflow/
COSEWIC 2002. COSEWIC assessment and update status report on the wild hyacinth Camassia scilloides in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. vi + 14 pp.
Crabtree, T. 2008. Tennessee Natural Heritage Program Rare Plant List. Division of Natural Areas, Tennessee Department of Environment and Conservation, Nashville, TN. 46 pp.
Environment Canada. 2006. Great Lakes Fact Sheet: The Rise of the Double-crested Cormorant on the Great Lakes: Winning the War Against Contaminants. On-line document, http://www.on.ec.gc.ca/wildlife/factsheets/fs_cormorants-e.html
Franken, K. M., J. M. Coons, H. R. Owen, E. L. Smith, and J. E. Ebinger. 2009.
Influence of Reproductive and Environmental Factors on Population Size of Wild Hyacinth [Camassia angusta (Engelm. and A. Gray) Blank. (Liliaceae)], an Illinois Endangered Species. Castanea 74(2).
Garibaldi, A. and N. Turner. 2004. Cultural keystone species: implications for ecological conservation and restoration. Ecology and Society 9(3): 1. [online] URL: http://www.ecologyandsociety.org/vol9/iss3/art1/
Gleason, H.A., and A. Cronquist. 1991. Manual of Vascular Plants of Northeastern United States and Adjacent Canada, 2nd ed. New York: The New York Botanical Gardens.
Gould, F. W. 1942. A systematic treatment of the genus Camassia Lindl. American Midland Naturalist 28(3).
Horvath, D. J., Blessman, G., Flood, R. M. 2001. Propagation protocol for production of container Camassia scilloides (Raf.) Cory plants (1+0 container plugs); Illinois Department of Natural Resources - Mason State Nursery, Topeka, Illinois. In: Native Plant Network. URL: http://www.nativeplantnetwork.org
Jalava, J.V. and P. Mansur. 2008a. National Recovery Strategy for Carolinian Woodlands and Associated Species at Risk, Phase II: Part 1 – Implementation. Draft 5, September 30, 2008. Carolinian Canada Coalition, London, Ontario. vii + 124pp.
Jalava, J.V., P.L. Wilson and R.A. Jones. 2008b. COSEWIC-designated Plant Species at Risk Inventory, Point Pelee National Park, including Sturgeon Creek Administrative Centre and Middle Island, 2007, Volume 1: Summary Report & Volume 2: Managed Area Element Status Assessments. Prepared for Parks Canada Agency, Point Pelee National Park Park, Leamington, Ontario. Vol. 1 vii + 126 pp., Vol. 2 ii + 103 pp.
Jalava, J.V., J.D. Ambrose and N. S. May. 2009. National Recovery Strategy for Carolinian Woodlands and Associated Species at Risk: Phase I. Draft 10 – March 31, 2009. Carolinian Canada Coalition and Ontario Ministry of Natural Resources, London, Ontario. viii + 75 pp.
Lee, H. 2012. Personal communications. SELC Master Tables (Draft update to Lee, H., W. Bakowsky, J. Riley, J. Bowles, M. Puddister, P. Uhlig and S. McMurray. 1998. Ecological Land Classification for Southern Ontario: First Approximation and Its Application. SCSS Field Guide FG-02. Ontario Ministry of Natural Resources. 225 pp.). Microsoft Excel Spreadsheet provided electronically to the author. Ecological Land Classification Ecologist, Ontario Ministry of Natural Resources, London, Ontario.
Lee, H., W. Bakowsky, J. Riley, J. Bowles, M. Puddister, P. Uhlig and S. McMurray. 1998. Ecological Land Classification for Southern Ontario: First Approximation and Its Application. SCSS Field Guide FG-02. Ontario Ministry of Natural Resources. 225 pp.
MacGregor, R.L. 1966. Additions to the Kansas flora. Transactions of the Kansas Academy of Science. Vol. 53 (3) p. 327.
Macior, L. W. 1978. Pollination ecology of vernal angiosperms. Oikos 30: 452-460.
McFarlane, M.H. 2012. Personal communications with J. Jalava, February 2012, re: Pelee Island Wild Hyacinth populations. Conservation Biologist, Southwestern Ontario, The Nature Conservancy of Canada, London, ON.
McKay, V.M. 2009. Personal communications with J. Jalava, March 2009, re: Middle Island Wild Hyacinth population surveys. Species At Risk Biologist, Parks Canada Agency, Point Pelee National Park.
NatureServe. 2006, 2012. NatureServe Explorer: An online encyclopedia of life [web application]. Version 6.1. NatureServe, Arlington, Virginia. Available http://www.natureserve.org/explorer. (Accessed: November 10, 2006 and February 2012).
NHIC (Natural Heritage Information Centre). 2006, 2009, 2012. Species Lists, Element Occurrence and Natural Areas databases and publications. Natural Heritage Information Centre, Ontario Ministry of Natural Resources, Peterborough, Ontario. Electronic databases.
North - South Environmental Inc. 2004. Vegetation communities and significant vascular plant species of Middle Island, Lake Erie. Prepared for Point Pelee National Park.
Oldham, M.J. 1990. COSEWIC status report on the wild hyacinth Camassia scilloides in Canada. Committee on the Status of Endangered Wildlife in Canada. Ottawa. 1-14 pp.
Ontario Nature. 2006. Stone Road Alvar Nature Reserve. On-line document, URL: http://www.ontarionature.org/enviroandcons/reserves/res_stone_road_alvar.html
Ontario Parks. 2005. Fish Point and Lighthouse Point Park Management Plan. Ontario Parks, Peterborough, Ontario. 25 pp.
PCA (Parks Canada Agency). 2011a. Detailed Assessment for the Wild Hyacinth (Camassia scilloides) in Point Pelee National Park (mainland). Species at Risk Detailed Assessments. Parks Canada Agency. Ottawa. 2 pp.
PCA (Parks Canada Agency). 2011b. Draft Managed Area Element Status Assessment – Middle Island (Wild Hyacinth). Parks Canada Agency, Ottawa. 8 pp.
Richards, N.R., A.G. Caldwell, and F. F. Morwick. 1949. Soil Survey of Essex County. Report No. 11 of the Ontario Soil Survey, Guelph, Ontario. 85 pp. + folded map.
Stevens, M., D.C. Darris and S.M. Lambert. 2001. Ethnobotany, Culture, Management and Use of Common Camas (Camassia quamash). Native Plants Journal 2(1) 47-53 (Spring 2001).
UTLBJWC (University of Texas Lady Bird Johnson Wildflower Center). 2012. Wild Hyacinth. http://www.wildflower.org/plants/result.php?id_plant=CASC5, accessed February 2012.
USDA (United States Department of Agriculture). 2012. Plants Profile: Camassia scilloides. On-line document, http://plants.usda.gov/java/profile?symbol=CASC5.
USFWS (United States Fish and Wildlife Service). 2012. The Migratory Bird Program – Conserving America’s Birds. http://www.fws.gov/migratorybirds/, accessed February 2012.
WDNR (Wisconsin Department of Natural Resources). 2006. http://www.dnr.state.wi.us/org/land/er/factsheets/plants/Hyacin.htm
Waldron, G. 2012. Personal communication with J. Ambrose, March 2012. Ecological consultant, Essex, Ontario.
Woodliffe, P.A. 2009, 2011. Personal communications with J. Jalava, February – March 2009, and review comments for a draft of this report in 2011. District Ecologist, OMNR Aylmer District, Chatham Area Office, Chatham, Ontario.
Recovery strategy development team members
Earlier drafts of this recovery strategy were developed by Jarmo Jalava in 2007 and 2009, with the assistance of the following Recovery Team members:
| Name | Affiliation and location |
|---|---|
| Roxanne St. Martin (Co-chair) | Ontario Ministry of Natural Resources |
| Michelle Kanter (Co-chair) | Carolinian Canada Coalition |
| John Ambrose | Botanical Consultant |
| Dawn Bazely | York University |
| Jane Bowles | University of Western Ontario |
| Barb Boysen | Ontario Ministry of Natural Resources |
| Dawn Burke | Ontario Ministry of Natural Resources |
| Peter Carson | Private Consultant / Ontario Nature |
| Ken Elliott | Ontario Ministry of Natural Resources |
| Mary Gartshore | Private Consultant |
| Ron Gould | Ontario Ministry of Natural Resources |
| Karen Hartley | Ontario Ministry of Natural Resources |
| Steve Hounsell | Ontario Power Generation |
| Donald Kirk | Ontario Ministry of Natural Resources |
| Daniel Kraus | Nature Conservancy of Canada |
| Nikki May | Carolinian Canada Coalition |
| Gordon Nelson | Carolinian Canada Coaltion / University of Waterloo |
| Michael Oldham | Ontario Ministry of Natural Resources |
| Bernie Solymar | Private Consultant |
| Tara Tchir | Upper Thames River Conservation Authority |
| Allen Woodliffe | Ontario Ministry of Natural Resources (formerly) |
Appendix 1: Considerations for monitoring wild hyacinth
Measures of the success of the recovery effort will form part of the regular monitoring program. Measures should include long-term trends in the size and number of extant sites (area of occupancy and area of extent), site quality (measured through a habitat suitability index) and population trends and projections determined through regular population counts. A scoring system should be developed that will allow for quantitative comparisons between Wild Hyacinth populations and factors affecting the quality and extent of its woodland habitat.
Monitoring may be undertaken at varying levels of intensity in the future depending on the current threat level, size and quality of each site. The following criteria are based in part on monitoring methods recommended in Bickerton (2003) and NatureServe (2006):
- A less-intensive level may be undertaken by volunteers or landowners annually or biannually. Performance measures will include the presence or absence of Wild Hyacinth and an approximate population count, a coarse numerical assessment of threats, and qualitative assessment of changes to habitat quality and threats.
- A more intensive level will involve demographic monitoring of Wild Hyacinth population trends based on life stages, seedling-establishment, mortality and other factors. Intensive monitoring may be considered for critical sites with a high-level of threat, sites for which qualified staff are available to conduct annual monitoring, and any re-introduction sites. Populations should be monitored to assess stability, note recruitment, document longevity of individuals and the yearly reproductive output of individual plants, as well as the nature and extent of impact on populations by threats (e.g., cormorant colonies, invasive species).
Footnotes
- footnote[1] Back to paragraph Other sources provide the following: Wild Hyacinth is described as: "common" in Missouri (CCM 2006), and it occurs in 22 counties in Kansas, 26 counties in Tennessee, 30 counties in eastern Texas and 5 counties in Alabama (USDA 2012). MacGregor (1966) considered it "common" in eastern Kansas, and it is not included in the Tennessee Natural Heritage Program Rare Plant list (Crabtree 2008). BONAP (2012) mapping suggests that at the county level it is only sporadically rare along the southern, eastern and northern edges of its range in North America. This suggests that, with the exception perhaps of Alabama, it is not of conservation concern in the states where its conservation status has not been ranked.
- footnote[2] Back to paragraph A population at Point Pelee National Park is believed to have originated from planted stock (NHIC 2006).
- footnote[3] Back to paragraph For the purpose of this recovery strategy, recovery habitat is considered historical but unoccupied habitat of Wild Hyacinth.